Difference between revisions of "Element"
| Line 16: | Line 16: | ||
|[[File:DiamondPicture.png|center|200px]] | |[[File:DiamondPicture.png|center|200px]] | ||
|- | |- | ||
| − | | style="height:20px; width:200px; text-align:center;" |[[Gold (Material)|Gold]] is an [[element]] made of [[Gold | + | | style="height:20px; width:200px; text-align:center;" |[[Gold (Material)|Gold]] is an [[element]] made of [[Gold]] [[atom]]s. |
| style="height:20px; width:200px; text-align:center;" |These lumps of [[Sulphur]] are made of only [[Sulphur]] [[atom]]s. | | style="height:20px; width:200px; text-align:center;" |These lumps of [[Sulphur]] are made of only [[Sulphur]] [[atom]]s. | ||
| style="height:20px; width:200px; text-align:center;" |This piece of [[Boron]] is made of only [[Boron]] [[atom]]s. | | style="height:20px; width:200px; text-align:center;" |This piece of [[Boron]] is made of only [[Boron]] [[atom]]s. | ||
Revision as of 10:39, 8 April 2019
Key Stage 3
Meaning
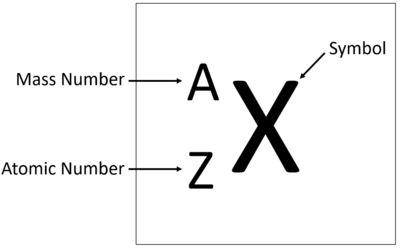
Elements are show on the Periodic Table with the Chemical Symbol, Atomic Number and Relative Atomic Mass.
An element is a material made of only one type of atom.
About Elements
- There are over 100 different elements and they are all shown on the Periodic Table.
- An element can be as little as one atom or could be trillions of identical atoms.
- If there are any other types of atom in an object it is not an element.
| Gold is an element made of Gold atoms. | These lumps of Sulphur are made of only Sulphur atoms. | This piece of Boron is made of only Boron atoms. | A diamond is made of only Carbon atoms making it pure carbon. |
- Elements can be represented in a particle diagram by showing only one colour or shade to represent that atoms.
| This is a solid element. An example would be gold. | This is a liquid element. An example would be Mercury. | This is a gaseous element. An example would be helium. | This gaseous element is made of molecules of the element. An example would be oxygen. |
Key Stage 4
Meaning
An element is a material made of atoms that all have the same atomic number.
About Elements
- Atoms of the same element all have the same chemical properties.
- There are several isotopes of each element which have slightly different physical properties such as density and melting point.