Difference between revisions of "Chemical Bond"
(→Examples) |
|||
| (10 intermediate revisions by one other user not shown) | |||
| Line 13: | Line 13: | ||
|+ Covalent Bonds | |+ Covalent Bonds | ||
|- | |- | ||
| − | |[[File:OxygenDotandCrossDiagram.png|center| | + | |[[File:OxygenDotandCrossDiagram.png|center|175px]] |
| − | |[[File:NitrogenDotandCrossDiagram.png|center| | + | |[[File:NitrogenDotandCrossDiagram.png|center|175px]] |
| − | |[[File:CarbonDioxideDotandCrossDiagram.png|center| | + | |[[File:CarbonDioxideDotandCrossDiagram.png|center|250px]] |
|- | |- | ||
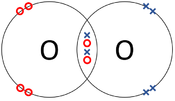
| − | | style="height:20px; width: | + | | style="height:20px; width:175px; text-align:center;" |The two [[Oxygen]] [[atom]]s each share two of their [[electron]]s with one another. |
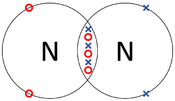
| − | | style="height:20px; width: | + | | style="height:20px; width:175px; text-align:center;" |The two [[Nitrogen]] [[atom]]s each share three of their [[electron]]s with one another. |
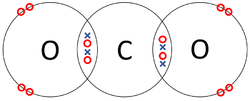
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |Each [[Oxygen]] shares two of its [[electron]]s with the [[Carbon]] [[atom]] while the [[Carbon]] [[atom]] shares two [[electron]]s with each [[Oxygen]] [[atom]]. |
|} | |} | ||
| Line 25: | Line 25: | ||
|+ Ionic Bonds | |+ Ionic Bonds | ||
|- | |- | ||
| − | |[[File: | + | |[[File:LithiumFluorideDotandCrossDiagram.png|center|200px]] |
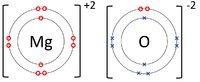
|[[File:MagnesiumOxideDotandCrossDiagram.png|center|200px]] | |[[File:MagnesiumOxideDotandCrossDiagram.png|center|200px]] | ||
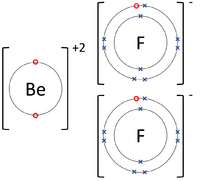
|[[File:BerylliumFluorideDotandCrossDiagram.png|center|200px]] | |[[File:BerylliumFluorideDotandCrossDiagram.png|center|200px]] | ||
|- | |- | ||
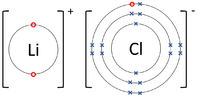
| − | | style="height:20px; width:200px; text-align:center;" | | + | | style="height:20px; width:200px; text-align:center;" |The [[Lithium]] [[atom]] donates an [[electron]] from its [[Outer Shell|outer shell]] to the [[Outer Shell|outer shell]] of the [[Fluorine]] [[atom]]. |
| − | | style="height:20px; width:200px; text-align:center;" | | + | | style="height:20px; width:200px; text-align:center;" |The [[Magnesium]] [[atom]] donates two [[electron]]s from its [[Outer Shell|outer shell]] to the [[Outer Shell|outer shell]] of the [[Oxygen]] [[atom]]. |
| − | | style="height:20px; width:200px; text-align:center;" | | + | | style="height:20px; width:200px; text-align:center;" |The [[Beryllium]] [[atom]] donates two [[electron]]s from its [[Outer Shell|outer shell]] to the [[Outer Shell|outer shells]] of each [[Fluorine]] [[atom]]. |
|} | |} | ||
{| class="wikitable" | {| class="wikitable" | ||
| − | |+ | + | |+ Metallic Bonds |
|- | |- | ||
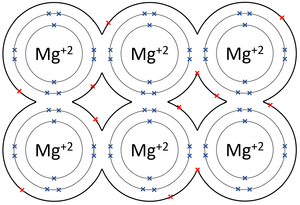
|[[File:MagnesiumMetallicBond.png|center|300px]] | |[[File:MagnesiumMetallicBond.png|center|300px]] | ||
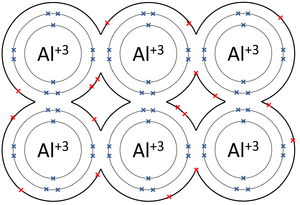
| − | |[[File: | + | |[[File:AluminiumMetallicBond.png|center|300px]] |
|- | |- | ||
| − | | style="height:20px; width:200px; text-align:center;" | | + | | style="height:20px; width:200px; text-align:center;" |The [[Outer Shell|outer shells]] of the [[Magnesium]] [[atom]]s overlap allowing the two [[electron]]s in each [[Outer Shell|outer shell]] to move freely between [[atom]]s. |
| − | | style="height:20px; width:200px; text-align:center;" | | + | | style="height:20px; width:200px; text-align:center;" |The [[Outer Shell|outer shells]] of the [[Aluminium]] [[atom]]s overlap allowing the three [[electron]]s in each [[Outer Shell|outer shell]] to move freely between [[atom]]s. |
|} | |} | ||
| + | |||
| + | ===Extra Information=== | ||
| + | {{#ev:youtube|https://www.youtube.com/watch?v=NgD9yHSJ29I}} | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Bonding, page 163, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Bonding, pages 47-8, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Bonding; covalent, pages 154, 156-7, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Bonding; covalent, pages 41-2, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Bonding; ionic, page 149, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Bonding; ionic, page 34, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Bonding; metallic, page 45, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Bonding; metallic, pages 160-1, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Chemical bond, pages 58-9, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | |||
| + | |||
| + | ====Edexcel==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Bond energies, page 136, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Bond energies, page 85, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Bond energies, pages 248-250, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bond energy, page 147, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bond energy, page 261, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding, page 186, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonding, page 42, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Bonding, pages 21, 23, 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Bonding, pages 51-53, 56-61, 65, 66, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Bonding, pages 84, 86, 88, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Bonding; covalent, page 23, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding; covalent, pages 184-185, 186, 192, 261, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonding; covalent, pages 40-41, 42, 48, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Bonding; covalent, pages 56-61, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding; giant covalent, page 192, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonding; giant covalent, page 48, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Bonding; ionic, page 21, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding; ionic, pages 178-179, 192, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Bonding; ionic, pages 51-53, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Bonding; metallic, page 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding; metallic, pages 190, 192, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonding; metallic, pages 46, 48, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Bonding; metallic, pages 65, 66, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonding; models, pages 192-193, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonding; models, pages 48-49, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonds; breaking and making, page 146, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonds; breaking and making, page 260, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonds; covalent, page 147, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Bonds; energy calculations, page 147, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Bonds; energy calculations, page 261, GCSE Combined Science, Pearson Edexcel ''] | ||
Latest revision as of 00:57, 3 November 2019
Contents
Key Stage 4
Meaning
A chemical bond is a force of attraction holding the atoms inside a molecule together.
About Chemical Bonds
There are three types of chemical bond you should know:
- Covalent Bonds - In which atoms share electrons with one another.
- Ionic Bonds - In which electrons are transferred from one atom to another.
- Metallic Bonds - In which some electrons move freely between atoms creating lattice of positively charged ions surrounded by a sea of delocalised electrons (free electrons).
Examples
| The two Oxygen atoms each share two of their electrons with one another. | The two Nitrogen atoms each share three of their electrons with one another. | Each Oxygen shares two of its electrons with the Carbon atom while the Carbon atom shares two electrons with each Oxygen atom. |
| The Lithium atom donates an electron from its outer shell to the outer shell of the Fluorine atom. | The Magnesium atom donates two electrons from its outer shell to the outer shell of the Oxygen atom. | The Beryllium atom donates two electrons from its outer shell to the outer shells of each Fluorine atom. |
| The outer shells of the Magnesium atoms overlap allowing the two electrons in each outer shell to move freely between atoms. | The outer shells of the Aluminium atoms overlap allowing the three electrons in each outer shell to move freely between atoms. |
Extra Information
References
AQA
- Bonding, page 163, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding, pages 47-8, GCSE Chemistry, Hodder, AQA
- Bonding; covalent, pages 154, 156-7, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding; covalent, pages 41-2, GCSE Chemistry, Hodder, AQA
- Bonding; ionic, page 149, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding; ionic, page 34, GCSE Chemistry, Hodder, AQA
- Bonding; metallic, page 45, GCSE Chemistry, Hodder, AQA
- Bonding; metallic, pages 160-1, GCSE Combined Science Trilogy 1, Hodder, AQA
- Chemical bond, pages 58-9, GCSE Chemistry; Student Book, Collins, AQA
Edexcel
- Bond energies, page 136, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Bond energies, page 85, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bond energies, pages 248-250, GCSE Chemistry, CGP, Edexcel
- Bond energy, page 147, GCSE Chemistry, Pearson, Edexcel
- Bond energy, page 261, GCSE Combined Science, Pearson Edexcel
- Bonding, page 186, GCSE Combined Science, Pearson Edexcel
- Bonding, page 42, GCSE Chemistry, Pearson, Edexcel
- Bonding, pages 21, 23, 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding, pages 51-53, 56-61, 65, 66, GCSE Chemistry, CGP, Edexcel
- Bonding, pages 84, 86, 88, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Bonding; covalent, page 23, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; covalent, pages 184-185, 186, 192, 261, GCSE Combined Science, Pearson Edexcel
- Bonding; covalent, pages 40-41, 42, 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; covalent, pages 56-61, GCSE Chemistry, CGP, Edexcel
- Bonding; giant covalent, page 192, GCSE Combined Science, Pearson Edexcel
- Bonding; giant covalent, page 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; ionic, page 21, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; ionic, pages 178-179, 192, GCSE Combined Science, Pearson Edexcel
- Bonding; ionic, pages 51-53, GCSE Chemistry, CGP, Edexcel
- Bonding; metallic, page 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; metallic, pages 190, 192, GCSE Combined Science, Pearson Edexcel
- Bonding; metallic, pages 46, 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; metallic, pages 65, 66, GCSE Chemistry, CGP, Edexcel
- Bonding; models, pages 192-193, GCSE Combined Science, Pearson Edexcel
- Bonding; models, pages 48-49, GCSE Chemistry, Pearson, Edexcel
- Bonds; breaking and making, page 146, GCSE Chemistry, Pearson, Edexcel
- Bonds; breaking and making, page 260, GCSE Combined Science, Pearson Edexcel
- Bonds; covalent, page 147, GCSE Chemistry, Pearson, Edexcel
- Bonds; energy calculations, page 147, GCSE Chemistry, Pearson, Edexcel
- Bonds; energy calculations, page 261, GCSE Combined Science, Pearson Edexcel