Difference between revisions of "Group 1"
(→Chemical Properties) |
(→Chemical Properties) |
||
| Line 31: | Line 31: | ||
The [[reactivity]] increases as you go down the group because: | The [[reactivity]] increases as you go down the group because: | ||
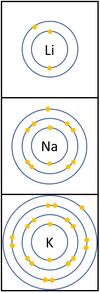
| − | *The outer [[electron]] is further away from the [[Atomic Nucleus|nucleus]] with each additional [[Electron Orbital|electron shell]] making the [[force]] of [[ | + | *The outer [[electron]] is further away from the [[Atomic Nucleus|nucleus]] with each additional [[Electron Orbital|electron shell]] making the [[force]] of [[attract]]ion weaker. |
*Even though the [[charge]] of the [[Atomic Nucleus|nucleus]] increases the outer [[electron]] is shielded from most of the [[Positive Charge|positive charge]] of the [[Atomic Nucleus|nucleus]] by [[electron]]s in the [[Inner Shell|inner shells]]. | *Even though the [[charge]] of the [[Atomic Nucleus|nucleus]] increases the outer [[electron]] is shielded from most of the [[Positive Charge|positive charge]] of the [[Atomic Nucleus|nucleus]] by [[electron]]s in the [[Inner Shell|inner shells]]. | ||
|} | |} | ||
Revision as of 18:06, 26 November 2018
Contents
Key Stage 4
Meaning
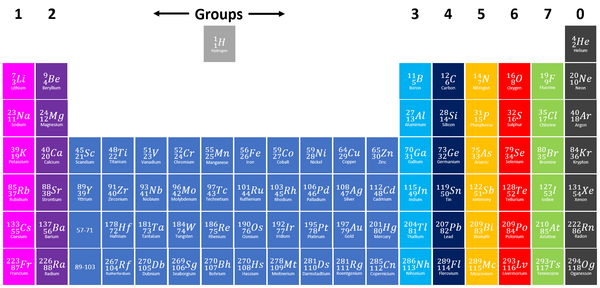
Group 1 elements, also known as Alkali Metals on the Periodic Table are the elements which have only one electron in their outer shell.
| Group 1 elements are shown in light purple at the far left of the Periodic Table. NB: Hydrogen is often shown in Group 1 due to having a single electron in the outer shell however it is not considered an Alkali Metal because it has very few properties in common with Alkali Metals. |
About the Alkali Metals
- The Alkali Metals have similar chemical properties because they all have one electron on their outer shell.
- Alkali Metals all produce ions with a +1 relative charge because they lose an electron in chemical reactions.
The Alkali Metals in order from least reactive to most reactive are:
Chemical Properties
- Alkali Metals are all highly reactive and will oxidise quickly in the presence of Oxygen.
- Alkali Metals all react strongly with water to produce metal hydroxides and Hydrogen gas.
- Alkali Metals all produce strong alkalis.
| In a chemical reaction the electron in the outer shell is lost.
The reactivity increases as you go down the group because:
|
Physical Properties
- Alkali Metals have a low density compared to other metals.
- Alkali Metals are all solid at room temperature but have a low melting point compared to other metals.
- Alkali Metals are soft and can be easily cut.
- Alkali Metals all appear shiny (before they oxidise).