Difference between revisions of "Nuclear Model"
| Line 38: | Line 38: | ||
:[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Nuclear model, pages 22, 23, 42, 43, GCSE Chemistry, CGP, AQA ''] | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Nuclear model, pages 22, 23, 42, 43, GCSE Chemistry, CGP, AQA ''] | ||
:[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Nuclear model, pages 22, 23, 42, 43, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Nuclear model, pages 22, 23, 42, 43, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | ||
| + | |||
| + | ====Edexcel==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Nuclear model, pages 32-34, GCSE Chemistry, CGP, Edexcel ''] | ||
Latest revision as of 18:38, 23 November 2019
Contents
Key Stage 4
Meaning
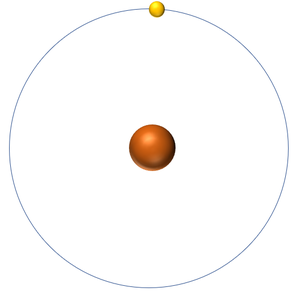
The Nuclear Model is a model of the atom which suggests there is a positively charged nucleus in the centre of an atom with electrons orbiting around the nucleus.
About the Nuclear Model
- The Nuclear Model was first proposed by Ernest Rutherford after he successfully disproved the Plum Pudding Model of the atom using his alpha scattering experiment.
- In the Nuclear Model most of the atom is empty space. The majority of the mass of the atom is concentrated in a small central nucleus. This central nucleus has a strong positive charge while negatively charged electrons orbit the nucleus.
Evidence for the Nuclear Model
- In Rutherford's Alpha Scattering Experiment several observations about the path of alpha particles through a thin sheet of Gold foil that led to the development of the Nuclear Model:
| Observation | Conclusion |
| Most of the alpha particles pass straight through the foil. | The atom must be mostly empty space. |
| Some of the alpha particles were deflected by a small angle. | The mass of the atom must be concentrated in an extremely small volume in the centre. |
| A very small number of alpha particles came back in the direction of the detector. (Deflected more than 90°.) | The centre of an atom must have a strong positive charge.
The electrons must not be in the centre of the atom, they must be orbiting the nucleus. |
References
AQA
- Nuclear model (atoms), pages 109, 110, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Nuclear model (atoms), pages 121, 122, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Nuclear model (of the atom), pages 104, 197, GCSE Combined Science; The Revision Guide, CGP, AQA
- Nuclear model of the atom, pages 91-2, GCSE Physics, Hodder, AQA
- Nuclear model, page 19, GCSE Chemistry; The Revision Guide, CGP, AQA
- Nuclear model, page 43, GCSE Physics; The Revision Guide, CGP, AQA
- Nuclear model, pages 22, 23, 42, 43, GCSE Chemistry, CGP, AQA
- Nuclear model, pages 22, 23, 42, 43, GCSE Combined Science Trilogy; Chemistry, CGP, AQA