Difference between revisions of "Chemical Formula"
(→About Chemical Formulae) |
|||
| Line 63: | Line 63: | ||
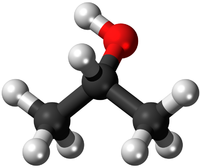
| style="height:20px; width:200px; text-align:center;" |[[Isopropyl Alcohol]] or [[Propan-2-ol]] has 3 [[Carbon]] [[atom]]s and 8 [[Hydrogen]] [[atom]]s and 1 [[Oxygen]] atom. This has the same '''chemical formula''' as [[Propan-1-ol]] but a different shape. This may be written with the [[Structural Formula|structural formula]] '''CH<sub>3</sub>CHOHCH<sub>3</sub>''' to show where the OH group is attached. | | style="height:20px; width:200px; text-align:center;" |[[Isopropyl Alcohol]] or [[Propan-2-ol]] has 3 [[Carbon]] [[atom]]s and 8 [[Hydrogen]] [[atom]]s and 1 [[Oxygen]] atom. This has the same '''chemical formula''' as [[Propan-1-ol]] but a different shape. This may be written with the [[Structural Formula|structural formula]] '''CH<sub>3</sub>CHOHCH<sub>3</sub>''' to show where the OH group is attached. | ||
|} | |} | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Chemical formulae, pages 36-37, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 13:11, 1 December 2019
Contents
Key Stage 3
Meaning
A Chemical Formula is a simple way to show the number and type of atoms in a chemical.
About Chemical Formulae
- Chemical Formulae show which elements are in a molecule.
- The number of each element in a molecule is shown with a number written just after and lower than the element.
Examples
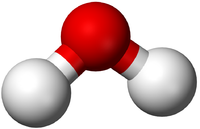
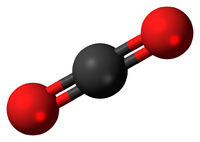
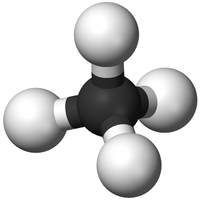
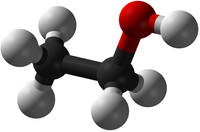
| H2O | CO2 | CH4 | C2H5OH |
| Water has 2 Hydrogen atoms and 1 Oxygen atom. | Carbon Dioxide has 1 Carbon atom and 2 Oxygen atoms. | Methane has 1 Carbon atom and 4 Hydrogen atoms. | Ethanol has 2 Carbon atoms, 6 Hydrogen atoms and 1 Oxygen atom. |
Key Stage 4
Meaning
A Chemical Formula is a way to show the numbers of atoms of each element in a chemical.
About Chemical Formulae
- Chemical Formulae show which elements are in a molecule.
- The number of each element in a molecule is shown with subscript after that element.
- Some chemical formulae have brackets which indicate that the elements in the bracket are multiplied by the number after the bracket.
- MgBr2: Magnesium Bromide with 1 Magnesium atom and 2 Bromine atoms.
- CO2: Carbon Dioxide with 1 Carbon atom and 2 Oxygen atoms.
- Li2O: Lithium Oxide with 2 Lithium atoms and 1 Oxygen atom.
- Al(OH)3: Aluminium Hydroxide with 1 Aluminium atom, with 3 lots of OH; 3 Oxygen atoms and 3 Hydrogen atoms.
- Al2(CO3)3: Aluminium Carbonate with 2 Aluminium atoms, and 3 lots of CO3; 3 Carbon atoms and 9 Oxygen atoms.
Some chemical formulae are written with the elements at specific points, not all grouped together, to show where different elements are bonded. This is called a structural formula.
- CH3COOH: Ethanoic Acid with 2 Carbon atoms, 2 Oxygen atoms and 4 Hydrogen atoms. This shows that one Carbon is bonded to three Hydrogen atoms while the other Carbon is double bonded to one Oxygen and has a single bond with an Oxygen, which itself it bonded to a Hydrogen.
- CH3CH2COOH: Propanoic Acid with three Carbon atoms, two Oxygen atoms and 6 Hydrogen atoms.
Examples
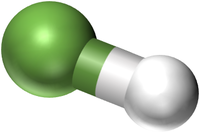
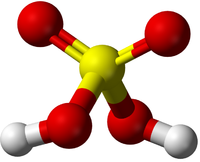
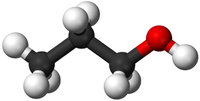
| HCl | H2SO4 | C3H8O | C3H8O |
| Hydrochloric Acid molecules have 1 Hydrogen atom and 1 Chlorine atom. | Sulphuric Acid molecules have 2 Hydrogen atoms, 1 Sulphur atom and 4 Oxygen atoms. | Propanol or Propan-1-ol has 3 Carbon atoms and 8 Hydrogen atoms and 1 Oxygen atom. This has the same chemical formula as Propan-2-ol but a different shape. This may be written with the structural formula CH3CH2CH2OH to show where the OH group is attached. | Isopropyl Alcohol or Propan-2-ol has 3 Carbon atoms and 8 Hydrogen atoms and 1 Oxygen atom. This has the same chemical formula as Propan-1-ol but a different shape. This may be written with the structural formula CH3CHOHCH3 to show where the OH group is attached. |