Difference between revisions of "Element"
| Line 108: | Line 108: | ||
:[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Elements; periodic table, pages 170-171, GCSE Combined Science, Pearson Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Elements; periodic table, pages 170-171, GCSE Combined Science, Pearson Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Elements; periodic table, pages 26-27, GCSE Chemistry, Pearson, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Elements; periodic table, pages 26-27, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Elements, pages 26, 52-53, 68-71, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 16:15, 5 December 2019
Contents
Key Stage 3
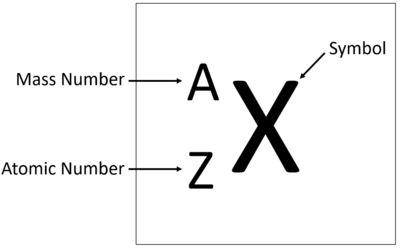
Elements are show on the Periodic Table with the Chemical Symbol, Atomic Number and Relative Atomic Mass.
Meaning
An element is a material made of only one type of atom.
About Elements
- There are over 100 different elements and they are all shown on the Periodic Table.
- An element can be as little as one atom or could be trillions of identical atoms.
- If there are any other types of atom in an object it is not an element.
| Gold is an element made of Gold atoms. | These lumps of Sulphur are made of only Sulphur atoms. | This piece of Boron is made of only Boron atoms. | A diamond is made of only Carbon atoms making it pure carbon. |
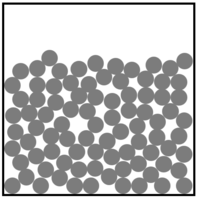
- Elements can be represented in a particle diagram by showing only one colour or shade to represent the atoms.
| This is a solid element. An example would be gold. | This is a liquid element. An example would be Mercury. | This is a gaseous element. An example would be helium. | This gaseous element is made of molecules of the element. An example would be oxygen. |
Key Stage 4

Elements are show on the Periodic Table with the Chemical Symbol, Atomic Number and Relative Atomic Mass.
Meaning
An element is a material made of atoms that all have the same atomic number.
About Elements
- Atoms of the same element all have the same chemical properties.
- There are several isotopes of each element which have slightly different physical properties such as density and melting point.
| Gold is an element made of Gold atoms. | These lumps of Sulphur are made of only Sulphur atoms. | This piece of Boron is made of only Boron atoms. | A diamond is made of only Carbon atoms making it pure carbon. |
- Elements can be represented in a particle diagram by showing only one colour or shade to represent the atoms.
| This is a solid element. An example would be gold. | This is a liquid element. An example would be Mercury. | This is a gaseous element. An example would be helium. | This gaseous element is made of molecules of the element. An example would be oxygen. |
References
AQA
- Element, pages 12-17, GCSE Chemistry; Student Book, Collins, AQA
- Element; boiling point, pages 34, 40-1, GCSE Chemistry; Student Book, Collins, AQA
- Element; formula, pages 62-3, 90-1, 252, GCSE Chemistry; Student Book, Collins, AQA
- Element; melting point, pages 34, 40-1, GCSE Chemistry; Student Book, Collins, AQA
- Element; reactivity, pages 38-9, 42-7, GCSE Chemistry; Student Book, Collins, AQA
- Elements, page 111, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Elements, page 123, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Elements, page 124, GCSE Combined Science Trilogy 1, Hodder, AQA
- Elements, page 13, GCSE Chemistry; The Revision Guide, CGP, AQA
- Elements, page 45, GCSE Physics; The Revision Guide, CGP, AQA
- Elements, page 9, GCSE Chemistry, Hodder, AQA
- Elements, page 97, GCSE Combined Science; The Revision Guide, CGP, AQA
- Elements, pages 24-26, GCSE Chemistry, CGP, AQA
- Elements, pages 24-26, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Elements, pages 4-5. 14-17, 38-43, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Elements; formula for, page 277, GCSE Chemistry, Hodder, AQA
- Elements; formulae for, page 244, GCSE Combined Science Trilogy 1, Hodder, AQA
- Elements; periodicity, pages 19,22-35, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Elements; reactions between, page 11, GCSE Chemistry, Hodder, AQA
- Elements; theories about, pages 145-6, GCSE Combined Science Trilogy 1, Hodder, AQA
Edexcel
- Elements, page 155, GCSE Physics, CGP, Edexcel
- Elements, page 18, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Elements, page 81, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Elements, page 90, GCSE Physics, Pearson Edexcel
- Elements, pages 148, 162, 186, 354, GCSE Combined Science, Pearson Edexcel
- Elements, pages 39-41, GCSE Chemistry, CGP, Edexcel
- Elements, pages 4, 18, 42, GCSE Chemistry, Pearson, Edexcel
- Elements; groups, page 173, GCSE Combined Science, Pearson Edexcel
- Elements; groups, page 29, GCSE Chemistry, Pearson, Edexcel
- Elements; periodic table, pages 170-171, GCSE Combined Science, Pearson Edexcel
- Elements; periodic table, pages 26-27, GCSE Chemistry, Pearson, Edexcel