Metal Ion
Revision as of 15:39, 13 January 2019 by NRJC (talk | contribs) (Created page with "==Key Stage 4== ===Meaning=== Metal Ions are positive ions found in ionic compounds and Giant Metallic Structure|giant metallic struc...")
Key Stage 4
Meaning
Metal Ions are positive ions found in ionic compounds and giant metallic structures.
About Metal Ions
- Metal ions are formed when metal elements lose their electrons to form positive ions.
The charge on a metal ion may be determined by the Group.
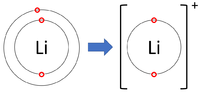
- Group 1 Elements all form +1 ions; Li+1, Na+1, K+1
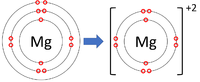
- Group 2 Elements all form +2 ions; Be+2, Mg+2, Ca+2
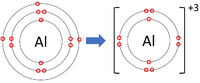
- Group 3 Elements all form +3 ions; Al+3
Transition Metal Elements can form different ions which are shown by Roman Numerals; Iron can form Fe(II) which is Fe+2 or Fe(III) is Fe+3, Manganese can form Mn(II) which is Mn+2 or Mn(IV) which is Mn+4.
Examples
| Lithium forms +1 ions. | Magnesium forms +2 ions. | Aluminium forms +3 ions. |