Difference between revisions of "Mixture"
| Line 106: | Line 106: | ||
:[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Mixtures; separation, page 10, GCSE Chemistry, Pearson, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Mixtures; separation, page 10, GCSE Chemistry, Pearson, Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Mixtures; separation, page 154, GCSE Combined Science, Pearson Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Mixtures; separation, page 154, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Mixtures, pages 26-29, 78, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Mixtures, pages 40-49, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Mixtures, pages 98-103, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
Revision as of 06:13, 15 December 2019
Contents
Key Stage 2
Meaning
A mixture is when two or more different substances are in the same place or are part of the same object.
About Mixtures
- Many materials are mixtures of different substances.
- Mixtures can usually be separated, but sometimes it is very difficult to do.
- Mixing is reversible change. Once two substances are mixed they can usually be separated again.
Examples
| Sea Water | Muddy Puddle | Beach |
| Seawater is mixture of salt and water. | This puddle is a mixture of mud and water. | The beach is a mixture of sand and pebbles. |
Key Stage 3
Meaning
A mixture is a substance which contains more than one type of chemical.
About Mixtures
- A mixture can be made of different elements or it can be made of different compounds.
- The different chemicals in a mixture are not chemically bonded with each other.
- Mixtures can be separated without a chemical reaction.
Examples
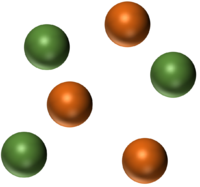
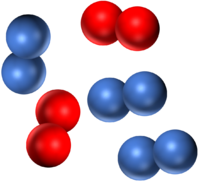
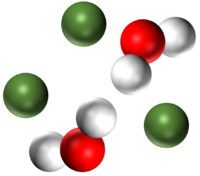
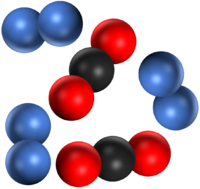
| A mixture of two different elements. | A mixture of two molecular elements. | A mixture of three different elements. |
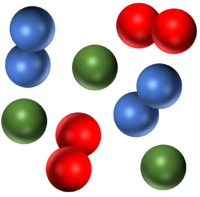
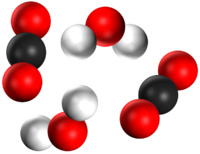
| A mixture of two compounds. | A mixture of a compound and an element. | A mixture of a compound and an element. |
Key Stage 4
Meaning
A mixture is a substance which contains more than one type of chemical.
About Mixtures
- Mixtures can be made of different elements, compounds or both.
- A mixture of metal elements is called an alloy.
- Mixtures can be separated by non-chemical processes.
Formulations
- A useful mixture may be referred to as a formulation. This is a mixture which combines the useful properties of several chemicals in the mixture.
Some examples of formulations are:
- Surface Cleaner - Contains bleach to kill bacteria, a fragrance to give it a pleasant smell and a surfactant to emulsify oils.
- Paint - A dye to provide colour and a solvent for it to be spread over a surface.
- Heartburn Relief - A base to neutralise stomach acid, alginic acid to form a layer to protect the lining of the stomach and oesophagus.
References
AQA
- Mixture, pages 12, 18-19, 262-5, GCSE Chemistry; Student Book, Collins, AQA
- Mixture; separation, pages 18-9, GCSE Chemistry; Student Book, Collins, AQA
- Mixtures, page 16-18, GCSE Chemistry; The Revision Guide, CGP, AQA
- Mixtures, pages 100-103, 150, GCSE Combined Science; The Revision Guide, CGP, AQA
- Mixtures, pages 136-7, GCSE Combined Science Trilogy 1, Hodder, AQA
- Mixtures, pages 155, GCSE Combined Science Trilogy 2, Hodder, AQA
- Mixtures, pages 21-2, 203, GCSE Chemistry, Hodder, AQA
- Mixtures, pages 34-41, GCSE Chemistry, CGP, AQA
- Mixtures, pages 34-41, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Mixtures, pages 8-9, 148-149, 180-181, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Mixtures; formulations, page 204, GCSE Chemistry, Hodder, AQA
- Mixtures; Formulations, pages 156, GCSE Combined Science Trilogy 2, Hodder, AQA
- Mixtures; Melting and boiling points, pages 155-6, GCSE Combined Science Trilogy 2, Hodder, AQA
- Mixtures; melting and boiling points, pages 203-4, GCSE Chemistry, Hodder, AQA
- Mixtures; rock salt, pages 25, GCSE Chemistry, Hodder, AQA
- Mixtures; separation of, pages 137-40, GCSE Combined Science Trilogy 1, Hodder, AQA
- Mixtures; separation of, pages 22-5, GCSE Chemistry, Hodder, AQA
- Mixtures; uses of, pages 203-4, GCSE Chemistry, Hodder, AQA
Edexcel
- Mixtures, page 36, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Mixtures, page 99, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Mixtures, pages 100, 101, GCSE Chemistry, CGP, Edexcel
- Mixtures, pages 148-149, GCSE Combined Science, Pearson Edexcel
- Mixtures, pages 4-5, GCSE Chemistry, Pearson, Edexcel
- Mixtures; separation, page 10, GCSE Chemistry, Pearson, Edexcel
- Mixtures; separation, page 154, GCSE Combined Science, Pearson Edexcel