Difference between revisions of "Silicon"
(→About Iodine) |
|||
| (9 intermediate revisions by 2 users not shown) | |||
| Line 3: | Line 3: | ||
[[Silicon]] is a [[solid]]. | [[Silicon]] is a [[solid]]. | ||
==Key Stage 3== | ==Key Stage 3== | ||
| + | [[File:SiliconSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Silicon]].]] | ||
| + | [[File:Si-28_WK.PNG|right|200px|thumb|A 2 dimensional representation of a [[Silicon]] [[atom]] with 14 [[proton]]s and 14 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 14 [[electron]]s orbiting the [[Atomic Nucleus|nucleus]].]] | ||
===Meaning=== | ===Meaning=== | ||
| − | |||
[[Silicon]] is a [[Group 4]] [[metalloid]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 14. | [[Silicon]] is a [[Group 4]] [[metalloid]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 14. | ||
===About Silicon=== | ===About Silicon=== | ||
| − | : [[Silicon]] has the [[Chemical Symbol|chemical symbol]] [[Silicon| | + | : [[Silicon]] has the [[Chemical Symbol|chemical symbol]] [[Silicon|Si]]. |
====Atomic Structure==== | ====Atomic Structure==== | ||
: [[Silicon]] as 14 [[proton]]s and 14 [[neutron]]s in its [[Atomic Nucleus|nucleus]] giving it an [[Atomic Number]] of 14 and an [[Relative Atomic Mass|atomic mass]] of 28. | : [[Silicon]] as 14 [[proton]]s and 14 [[neutron]]s in its [[Atomic Nucleus|nucleus]] giving it an [[Atomic Number]] of 14 and an [[Relative Atomic Mass|atomic mass]] of 28. | ||
| Line 14: | Line 15: | ||
====Properties==== | ====Properties==== | ||
: [[Silicon]] is a shiny [[solid]] at [[STP|room temperature]]. | : [[Silicon]] is a shiny [[solid]] at [[STP|room temperature]]. | ||
| + | |||
==Key Stage 4== | ==Key Stage 4== | ||
| + | [[File:SiKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Silicon]].]] | ||
| + | [[File:Si-28_WK.PNG|right|200px|thumb|A 2 dimensional representation of the [[Bohr Model]] of a [[Silicon]]-28 [[isotope]] with 14 [[proton]]s and 14 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 2 [[electron]]s in the first [[Electron Orbital|shell]], 8 in the second and 4 in the [[Outer Shell|outer shell]].]] | ||
===Meaning=== | ===Meaning=== | ||
[[Silicon]] is a [[Group 4]] [[metalloid]] [[element]], on the [[Periodic Table]], with 14 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | [[Silicon]] is a [[Group 4]] [[metalloid]] [[element]], on the [[Periodic Table]], with 14 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | ||
| − | ===About | + | ===About Silicon=== |
: [[Silicon]] has the [[Chemical Symbol|chemical symbol]] [[Silicon|Si]]. | : [[Silicon]] has the [[Chemical Symbol|chemical symbol]] [[Silicon|Si]]. | ||
====Atomic Structure==== | ====Atomic Structure==== | ||
| Line 25: | Line 29: | ||
: [[Silicon]] is a [[metalloid]] [[element]] so in some conditions it is an [[Electrical Conductor|electrical conductor]]. | : [[Silicon]] is a [[metalloid]] [[element]] so in some conditions it is an [[Electrical Conductor|electrical conductor]]. | ||
: [[Silicon]] is a shiny [[solid]] at [[STP|standard temperature and pressure]] and has a high [[Melting Point|melting point]]. | : [[Silicon]] is a shiny [[solid]] at [[STP|standard temperature and pressure]] and has a high [[Melting Point|melting point]]. | ||
| + | |||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Silicon, page 133, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Silicon, page 226, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158770/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158770&linkCode=as2&tag=nrjc-21&linkId=ec31595e720e1529e49876c3866fff6e ''Silicon, page 287, GCSE Physics; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Silicon; dioxide, pages 64, 76-7, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
Latest revision as of 14:00, 19 February 2021
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Silicon is a Group 4 metalloid element, on the Periodic Table, with an atomic number of 14.
About Silicon
- Silicon has the chemical symbol Si.
Atomic Structure
- Silicon as 14 protons and 14 neutrons in its nucleus giving it an Atomic Number of 14 and an atomic mass of 28.
- Silicon is in Period 3 of the Periodic Table because it has 3 electron shells.
Properties
- Silicon is a shiny solid at room temperature.
Key Stage 4
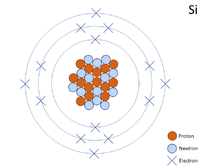
A 2 dimensional representation of the Bohr Model of a Silicon-28 isotope with 14 protons and 14 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second and 4 in the outer shell.
Meaning
Silicon is a Group 4 metalloid element, on the Periodic Table, with 14 protons in the nucleus.
About Silicon
- Silicon has the chemical symbol Si.
Atomic Structure
- The most stable isotope of Silicon has 14 neutrons in its nucleus giving it an atomic mass of 28.
- Silicon is in Period 3 of the Periodic Table because it has 3 electron shells.
Properties
- Silicon is a metalloid element so in some conditions it is an electrical conductor.
- Silicon is a shiny solid at standard temperature and pressure and has a high melting point.
