Difference between revisions of "Group 1"
| (17 intermediate revisions by 2 users not shown) | |||
| Line 11: | Line 11: | ||
===About the Alkali Metals=== | ===About the Alkali Metals=== | ||
| + | : Though [[Hydrogen]] has one [[electron]] in the [[Outer Shell|outer shell]] it does not behave the same as other [[element]]s in '''group 1''' so it is not considered to be an '''Alkali Metal'''. | ||
: The '''Alkali Metals''' have similar [[Chemical Property|chemical properties]] because they all have one [[electron]] on their [[Outer Shell|outer shell]]. | : The '''Alkali Metals''' have similar [[Chemical Property|chemical properties]] because they all have one [[electron]] on their [[Outer Shell|outer shell]]. | ||
: '''Alkali Metals''' all produce [[ion]]s with a +1 [[Relative Atomic Charge|relative charge]] because they lose an [[electron]] in [[Chemical Reaction|chemical reactions]]. | : '''Alkali Metals''' all produce [[ion]]s with a +1 [[Relative Atomic Charge|relative charge]] because they lose an [[electron]] in [[Chemical Reaction|chemical reactions]]. | ||
| Line 22: | Line 23: | ||
===Chemical Properties=== | ===Chemical Properties=== | ||
| − | : '''Alkali Metals''' are all highly [[Reactivity|reactive]] and will [[ | + | : '''Alkali Metals''' are all highly [[Reactivity|reactive]] and will [[Oxidation|oxidise]] quickly in the presence of [[Oxygen]]. |
: '''Alkali Metals''' all [[Chemical Reaction|react]] strongly with [[water]] to produce [[Metal Hydroxide|metal hydroxides]] and [[Hydrogen]] [[gas]]. | : '''Alkali Metals''' all [[Chemical Reaction|react]] strongly with [[water]] to produce [[Metal Hydroxide|metal hydroxides]] and [[Hydrogen]] [[gas]]. | ||
: '''Alkali Metals''' all produce strong [[alkali]]s. | : '''Alkali Metals''' all produce strong [[alkali]]s. | ||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| − | |[[File:Group1ElectronShells.png|center| | + | |[[File:Group1ElectronShells.png|center|100px]] |
| − | | style="height:20px; width: | + | |[[File:Group1ElectronShielding.png|center|100px]] |
| − | * | + | | style="height:20px; width:400px; text-align:left;" |In a [[Chemical Reaction|chemical reaction]] the [[electron]] in the [[Outer Shell|outer shell]] is lost. |
| + | |||
| + | The [[reactivity]] increases as you go down the group because: | ||
| + | *The outer [[electron]] is further away from the [[Atomic Nucleus|nucleus]] with each additional [[Electron Orbital|electron shell]] making the [[force]] of [[attract]]ion weaker. This makes it easier for an [[atom]] to lose it's outer [[electron]]. | ||
| + | *Even though the [[Electrical Charge|charge]] of the [[Atomic Nucleus|nucleus]] increases the outer [[electron]] is shielded from most of the [[Positive Charge|positive charge]] of the [[Atomic Nucleus|nucleus]] by [[electron]]s in the inner shells. | ||
|} | |} | ||
| Line 36: | Line 41: | ||
: '''Alkali Metals''' are all [[solid]] at [[Room Temperature|room temperature]] but have a low [[Melting Point|melting point]] compared to other [[metal]]s. | : '''Alkali Metals''' are all [[solid]] at [[Room Temperature|room temperature]] but have a low [[Melting Point|melting point]] compared to other [[metal]]s. | ||
: '''Alkali Metals''' are soft and can be easily cut. | : '''Alkali Metals''' are soft and can be easily cut. | ||
| − | : '''Alkali Metals''' all appear shiny (before they [[ | + | : '''Alkali Metals''' all appear shiny (before they [[oxidation|oxidise]]). |
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Alkali metals (Group 1), pages 14-15, GCSE Chemistry, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945598/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945598&linkCode=as2&tag=nrjc-21&linkId=ad276ad49df77ab4b40ab4fd0fe09664 ''Alkali metals, page 109, GCSE Combined Science; The Revision Guide, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d34 ''Alkali metals, page 24, GCSE Chemistry; The Revision Guide, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Alkali metals, pages 129-31, GCSE Combined Science Trilogy 1, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Alkali metals, pages 26-27, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Alkali metals, pages 56-58, GCSE Combined Science Trilogy; Chemistry, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Alkali metals, pages 56-59, 66, GCSE Chemistry, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Alkali metals, physical properties, page 162, GCSE Combined Science Trilogy 1, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Group 1 (alkali metals), pages 129-31, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Group 1 (alkali metals), pages 42-3, 46, 49, 42-3, 131, 133-5, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Group 1 (alkali metals); physical properties, page 162, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945598/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945598&linkCode=as2&tag=nrjc-21&linkId=ad276ad49df77ab4b40ab4fd0fe09971 ''Group 1, page 109, GCSE Combined Science; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d146 ''Group 1, page 24, GCSE Chemistry; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Group 1, pages 14-15, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Group 1, pages 26-27, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Group 1, pages 56-58, 66, GCSE Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Group 1, pages 56-58, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | ||
| + | |||
| + | ====Edexcel==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Alkali metals, page 123, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Alkali metals, page 73, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Alkali metals, pages 128-129, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Alkali metals, pages 242-243, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Alkali metals, pages 40, 49, 210-212, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Group 1, pages 20, 73, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Group 1, pages 40, 49, 210-212, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Group 1, pages 83, 123, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Alkali metals (Group 1 elements), pages 70, 71, 132-133, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Alkali metals, page 121, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Alkali metals, page 51, GCSE Chemistry; The Revision Guide, CGP, OCR Gateway ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Group 1 elements, pages 70, 71, 132-133, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Group 1, page 121, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Group 1, page 51, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
Latest revision as of 15:32, 11 December 2019
Contents
Key Stage 4
Meaning
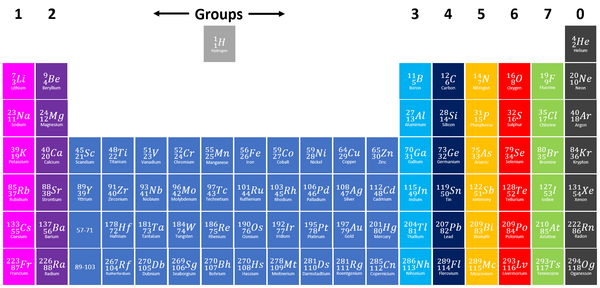
Group 1 elements, also known as Alkali Metals on the Periodic Table are the elements which have only one electron in their outer shell.
| Group 1 elements are shown in light purple at the far left of the Periodic Table. NB: Hydrogen is often shown in Group 1 due to having a single electron in the outer shell however it is not considered an Alkali Metal because it has very few properties in common with Alkali Metals. |
About the Alkali Metals
- Though Hydrogen has one electron in the outer shell it does not behave the same as other elements in group 1 so it is not considered to be an Alkali Metal.
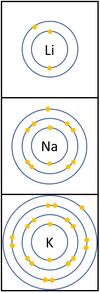
- The Alkali Metals have similar chemical properties because they all have one electron on their outer shell.
- Alkali Metals all produce ions with a +1 relative charge because they lose an electron in chemical reactions.
The Alkali Metals in order from least reactive to most reactive are:
Chemical Properties
- Alkali Metals are all highly reactive and will oxidise quickly in the presence of Oxygen.
- Alkali Metals all react strongly with water to produce metal hydroxides and Hydrogen gas.
- Alkali Metals all produce strong alkalis.
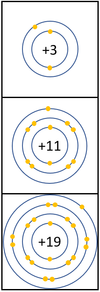
| In a chemical reaction the electron in the outer shell is lost.
The reactivity increases as you go down the group because:
|
Physical Properties
- Alkali Metals have a low density compared to other metals.
- Alkali Metals are all solid at room temperature but have a low melting point compared to other metals.
- Alkali Metals are soft and can be easily cut.
- Alkali Metals all appear shiny (before they oxidise).
References
AQA
- Alkali metals (Group 1), pages 14-15, GCSE Chemistry, Hodder, AQA
- Alkali metals, page 109, GCSE Combined Science; The Revision Guide, CGP, AQA
- Alkali metals, page 24, GCSE Chemistry; The Revision Guide, CGP, AQA
- Alkali metals, pages 129-31, GCSE Combined Science Trilogy 1, Hodder, AQA
- Alkali metals, pages 26-27, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Alkali metals, pages 56-58, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Alkali metals, pages 56-59, 66, GCSE Chemistry, CGP, AQA
- Alkali metals, physical properties, page 162, GCSE Combined Science Trilogy 1, Hodder, AQA
- Group 1 (alkali metals), pages 129-31, GCSE Combined Science Trilogy 1, Hodder, AQA
- Group 1 (alkali metals), pages 42-3, 46, 49, 42-3, 131, 133-5, GCSE Chemistry; Student Book, Collins, AQA
- Group 1 (alkali metals); physical properties, page 162, GCSE Combined Science Trilogy 1, Hodder, AQA
- Group 1, page 109, GCSE Combined Science; The Revision Guide, CGP, AQA
- Group 1, page 24, GCSE Chemistry; The Revision Guide, CGP, AQA
- Group 1, pages 14-15, GCSE Chemistry, Hodder, AQA
- Group 1, pages 26-27, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Group 1, pages 56-58, 66, GCSE Chemistry, CGP, AQA
- Group 1, pages 56-58, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
Edexcel
- Alkali metals, page 123, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Alkali metals, page 73, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Alkali metals, pages 128-129, GCSE Chemistry, Pearson, Edexcel
- Alkali metals, pages 242-243, GCSE Combined Science, Pearson Edexcel
- Alkali metals, pages 40, 49, 210-212, GCSE Chemistry, CGP, Edexcel
- Group 1, pages 20, 73, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Group 1, pages 40, 49, 210-212, GCSE Chemistry, CGP, Edexcel
- Group 1, pages 83, 123, GCSE Combined Science; The Revision Guide, CGP, Edexcel
OCR
- Alkali metals (Group 1 elements), pages 70, 71, 132-133, Gateway GCSE Chemistry, Oxford, OCR
- Alkali metals, page 121, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR
- Alkali metals, page 51, GCSE Chemistry; The Revision Guide, CGP, OCR Gateway
- Group 1 elements, pages 70, 71, 132-133, Gateway GCSE Chemistry, Oxford, OCR
- Group 1, page 121, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR
- Group 1, page 51, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR