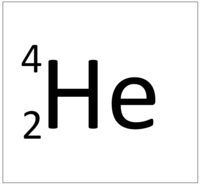Atom
Contents
Key Stage 3
Meaning
An atom is a very small particle made of protons, neutrons and electrons that can join with other atoms to make molecules.
About Atoms in The Dalton Model
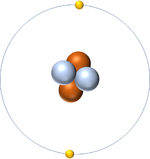
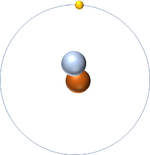
- In The Dalton Model atoms are shown as ball shaped particles. This makes it easier to draw diagrams of molecules.
| A picture of The Dalton Model of an atom. |
About Atoms beyond The Dalton Model
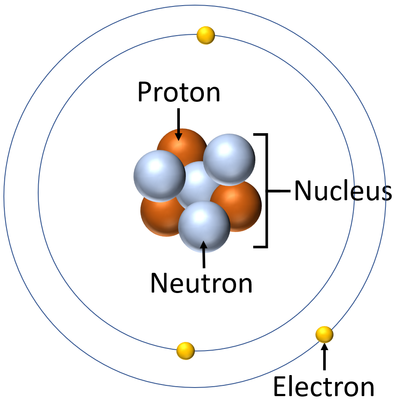
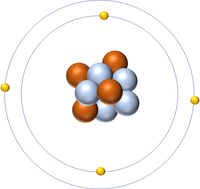
- Atoms are made of three smaller particles; the proton, neutron and electron.
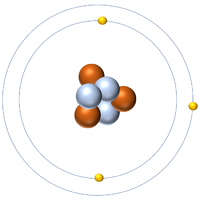
- Protons and neutrons are found in the nucleus at the centre of an atom. Electrons are found orbiting the nucleus in 'shells'.
| A diagram of an atom. |
- In an atom the number of electrons is always the same as the number of protons in the nucleus.
- Different atoms can have different numbers of protons and neutrons.
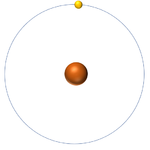
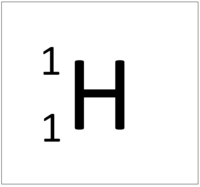
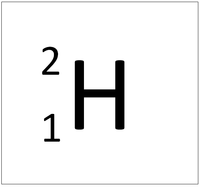
- The simplest atom is Hydrogen which has 1 proton and 1 electron and no neutrons.
Key Stage 4
Meaning
An atom is a very small particle made of protons, neutrons and electrons that can join with other atoms to make molecules.
About Atoms
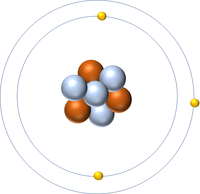
- Atoms consist of a small, central nucleus containing protons and neutrons surrounded by electrons orbiting the nucleus.
- The electrons orbit the nucleus in so called 'electron shells.
| A diagram of an atom. |
- In an atom there is always the same number of protons as electrons. If any electron is added or removed the atom becomes an ion.
| Hydrogen | Helium | Lithium | Beryllium |
| Hydrogen always has 1 proton. | Helium always has 2 protons. | Lithium always has 3 protons. | Beryllium always has 4 protons. |
- Atoms of the same element can have different numbers of neutrons so they can be different isotopes of the same element.
| Hydrogen-1 | Hydrogen-2 | Lithium-7 | Lithium-6 |
| Hydrogen always has 1 proton but in this case has no neutrons. | Hydrogen always has 1 proton but in this case also has a neutron. This isotope of Hydrogen is known as Deuterium. | Lithium always has 3 protons but in this case has 4 neutrons. | Lithium always has 3 protons but in this case has 3 neutrons. |
Properties of the Subatomic Particles
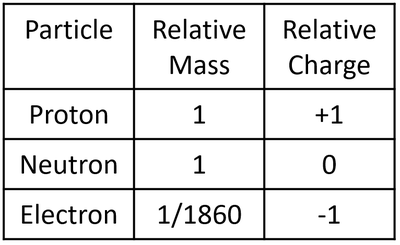
- The particles that make an atom all have slightly different properties. Since the particles are so small their mass and charge are extremely small numbers so, to make it easier, they are represented as 'relative' mass and 'relative' charge compared to a proton.
| A table showing the relative mass and relative charge of the proton, neutron and electron. |
History of Atoms
- The existence and structure of atoms was not always known.