Difference between revisions of "Hydrogen"
| Line 8: | Line 8: | ||
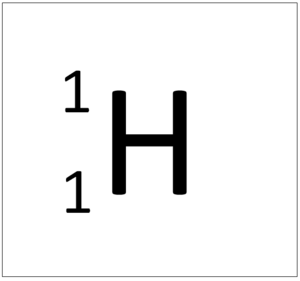
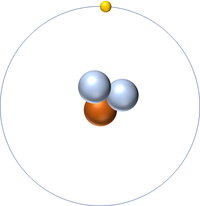
[[Hydrogen]] is a [[non-metal]] [[element]] with 1 [[proton]] in the [[Atomic Nucleus|nucleus]]. | [[Hydrogen]] is a [[non-metal]] [[element]] with 1 [[proton]] in the [[Atomic Nucleus|nucleus]]. | ||
===About Hydrogen=== | ===About Hydrogen=== | ||
| + | ====Molecular Structure==== | ||
: [[Hydrogen]] has the [[Chemical Formula|chemical formula]] [[Hydrogen|H<sub>2</sub>]]. | : [[Hydrogen]] has the [[Chemical Formula|chemical formula]] [[Hydrogen|H<sub>2</sub>]]. | ||
| + | : Two [[Hydrogen]] [[atom]]s join together to form a [[Hydrogen]] [[molecule]]. | ||
| + | ====Atomic Structure==== | ||
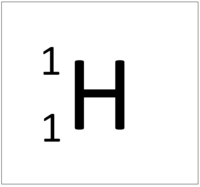
| + | : [[Hydrogen]] has 1 [[proton]] and 0 [[neutron]]s so it has an [[Relative Atomic Mass|atomic mass]] of 1. | ||
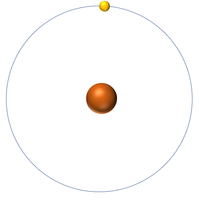
| + | : An [[atom]] of [[hydrogen]] has one [[electron]]. | ||
| + | : A [[Hydrogen]] [[ion]] has usually lost its only [[electron]] to become [[Positive Charge|positively charged]]. | ||
| + | ====Properties==== | ||
: [[Hydrogen]] is a [[gas]] at [[STP|room temperature]]. | : [[Hydrogen]] is a [[gas]] at [[STP|room temperature]]. | ||
: [[Hydrogen]] [[gas]] is less [[density|dense]] than [[air]]. | : [[Hydrogen]] [[gas]] is less [[density|dense]] than [[air]]. | ||
| − | : | + | : [[Hydrogen]] [[Chemical Reaction|reacts]] with [[Oxygen]] to form [[water]]. |
| − | + | ||
| − | ===Testing for Hydrogen=== | + | ====Testing for Hydrogen==== |
#Collect the [[gas]] in a [[Test Tube|test tube]]. | #Collect the [[gas]] in a [[Test Tube|test tube]]. | ||
#Place a lit [[Wooden Splint|splint]] over the mouth of the [[Test Tube|test tube]]. | #Place a lit [[Wooden Splint|splint]] over the mouth of the [[Test Tube|test tube]]. | ||
#If a 'squeaky pop' [[sound]] is made then the [[gas]] is [[Hydrogen]]. | #If a 'squeaky pop' [[sound]] is made then the [[gas]] is [[Hydrogen]]. | ||
| + | |||
==Key Stage 4== | ==Key Stage 4== | ||
===Meaning=== | ===Meaning=== | ||
[[Hydrogen]] is a [[non-metal]] [[element]] with 1 [[proton]] in the [[Atomic Nucleus|nucleus]]. | [[Hydrogen]] is a [[non-metal]] [[element]] with 1 [[proton]] in the [[Atomic Nucleus|nucleus]]. | ||
===About Hydrogen=== | ===About Hydrogen=== | ||
| + | ====Molecular Structure==== | ||
: [[Hydrogen]] has the [[Chemical Formula|chemical formula]] [[Hydrogen|H<sub>2</sub>]]. | : [[Hydrogen]] has the [[Chemical Formula|chemical formula]] [[Hydrogen|H<sub>2</sub>]]. | ||
| − | : [[ | + | : Two [[Iodine]] [[atom]]s join together in a [[Covalent Bond|covalent bond]]. |
| − | + | {| class="wikitable" | |
| − | : | + | |- |
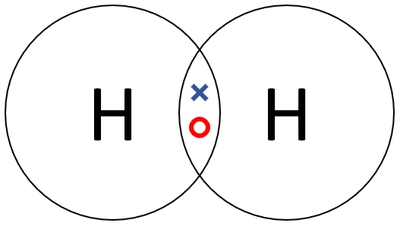
| − | + | |[[File:HydrogenDotandCrossDiagram.png|center|400px]] | |
| − | + | |- | |
| + | | style="height:20px; width:200px; text-align:center;" |A [[Dot and Cross Diagram|dot and cross diagram]] of a [[Hydrogen]] [[molecule]]. | ||
| + | |} | ||
| + | ====Atomic Structure==== | ||
| + | : The most common [[isotope]] of [[Hydrogen]] has 1 [[proton]] and 0 [[neutron]]s so it has an [[Relative Atomic Mass|atomic mass]] of 1. | ||
| + | |||
{| class="wikitable" | {| class="wikitable" | ||
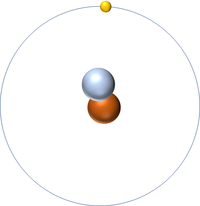
| style="height:20px; width:200px; text-align:center;" |[[Hydrogen]] | | style="height:20px; width:200px; text-align:center;" |[[Hydrogen]] | ||
| Line 44: | Line 58: | ||
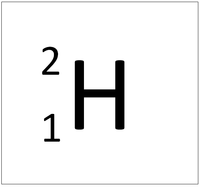
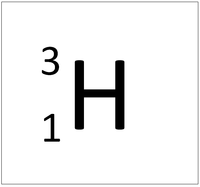
| style="height:20px; width:200px; text-align:center;" |[[Hydrogen]] always has 1 [[proton]] but in this [[isotope]] there is 2 [[neutron]]s. This [[isotope]] of [[Hydrogen]] is known as [[Tritium]]. | | style="height:20px; width:200px; text-align:center;" |[[Hydrogen]] always has 1 [[proton]] but in this [[isotope]] there is 2 [[neutron]]s. This [[isotope]] of [[Hydrogen]] is known as [[Tritium]]. | ||
|} | |} | ||
| + | : An [[atom]] of [[hydrogen]] has one [[electron]]. | ||
| + | : A [[Hydrogen]] [[ion]] has usually lost its only [[electron]] to become [[Positive Charge|positively charged]]. | ||
| + | ====Properties==== | ||
| + | : [[Hydrogen]] is a [[gas]] at [[STP|standard temperature and pressure]]. | ||
| + | : [[Hydrogen]] [[gas]] is less [[density|dense]] than [[air]]. | ||
| + | : [[Hydrogen]] [[Chemical Reaction|reacts]] with [[Oxygen]] to form [[water]]. | ||
| + | |||
===Testing for Hydrogen=== | ===Testing for Hydrogen=== | ||
#Collect the [[gas]] in a [[Test Tube|test tube]]. | #Collect the [[gas]] in a [[Test Tube|test tube]]. | ||
#Place a lit [[Wooden Splint|splint]] over the mouth of the [[Test Tube|test tube]]. | #Place a lit [[Wooden Splint|splint]] over the mouth of the [[Test Tube|test tube]]. | ||
#If a 'squeaky pop' [[sound]] is made then the [[gas]] is [[Hydrogen]]. | #If a 'squeaky pop' [[sound]] is made then the [[gas]] is [[Hydrogen]]. | ||
Revision as of 19:03, 31 March 2019
Contents
Key Stage 2
Meaning
Hydrogen is a gas that can catch fire easily.
Key Stage 3
Meaning
Hydrogen is a non-metal element with 1 proton in the nucleus.
About Hydrogen
Molecular Structure
Atomic Structure
- Hydrogen has 1 proton and 0 neutrons so it has an atomic mass of 1.
- An atom of hydrogen has one electron.
- A Hydrogen ion has usually lost its only electron to become positively charged.
Properties
- Hydrogen is a gas at room temperature.
- Hydrogen gas is less dense than air.
- Hydrogen reacts with Oxygen to form water.
Testing for Hydrogen
- Collect the gas in a test tube.
- Place a lit splint over the mouth of the test tube.
- If a 'squeaky pop' sound is made then the gas is Hydrogen.
Key Stage 4
Meaning
Hydrogen is a non-metal element with 1 proton in the nucleus.
About Hydrogen
Molecular Structure
- Hydrogen has the chemical formula H2.
- Two Iodine atoms join together in a covalent bond.
| A dot and cross diagram of a Hydrogen molecule. |
Atomic Structure
- The most common isotope of Hydrogen has 1 proton and 0 neutrons so it has an atomic mass of 1.
| Hydrogen | Deuterium | Tritium |
| Hydrogen always has 1 proton but isotope there are no neutrons. | Hydrogen always has 1 proton but in this isotope there is 1 neutron. This isotope of Hydrogen is known as Deuterium. | Hydrogen always has 1 proton but in this isotope there is 2 neutrons. This isotope of Hydrogen is known as Tritium. |
- An atom of hydrogen has one electron.
- A Hydrogen ion has usually lost its only electron to become positively charged.
Properties
- Hydrogen is a gas at standard temperature and pressure.
- Hydrogen gas is less dense than air.
- Hydrogen reacts with Oxygen to form water.