Difference between revisions of "Periodic Table"
| Line 55: | Line 55: | ||
===Meaning=== | ===Meaning=== | ||
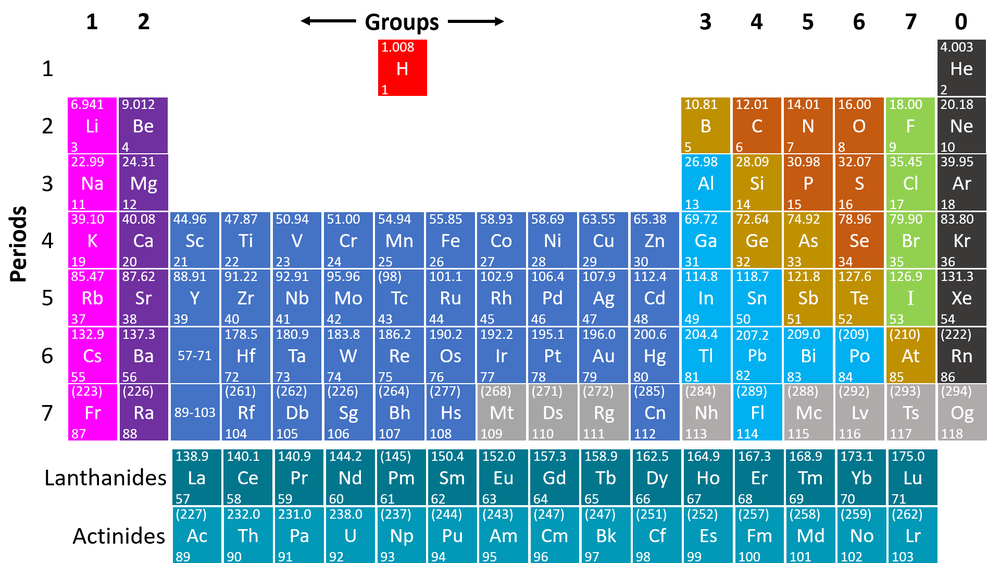
The [[Periodic Table]] is a chart listing all the known [[element]]s arranged in order of [[Atomic Number]] and in columns of [[element]]s with similar [[Property|properties]]. | The [[Periodic Table]] is a chart listing all the known [[element]]s arranged in order of [[Atomic Number]] and in columns of [[element]]s with similar [[Property|properties]]. | ||
| + | |||
| + | ===About The Periodic Table=== | ||
| + | : The modern [[Periodic Table]] has the [[element]]s arranged by [[Atomic Number]] and [[Chemical Property|chemical properties]]. | ||
| + | : [[Element]]s on the [[Periodic Table]] are written with their [[Chemical Symbol]]s and two numbers representing their [[Relative Atomic Mass]] and [[Atomic Number]]. | ||
| + | : The [[Relative Atomic Mass]]es on the [[Periodic Table]] are an [[Mean Average|average]] [[Relative Atomic Mass|atomic mass]] of different [[isotope]]s of the same [[element]]. | ||
| + | : The columns of the [[Periodic Table]] are called [[Group (Chemistry)|group]]s. | ||
| + | : The rows of the [[Periodic Table]] are called [[Period (Chemistry)|period]]s. | ||
{| class="wikitable" | {| class="wikitable" | ||
Revision as of 13:00, 5 December 2018
Contents
Key Stage 3
Meaning
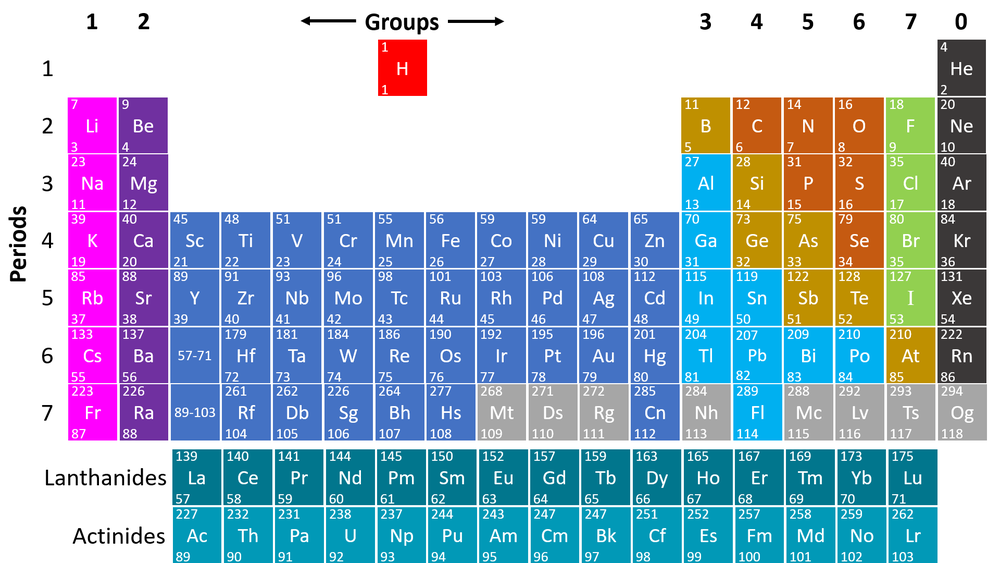
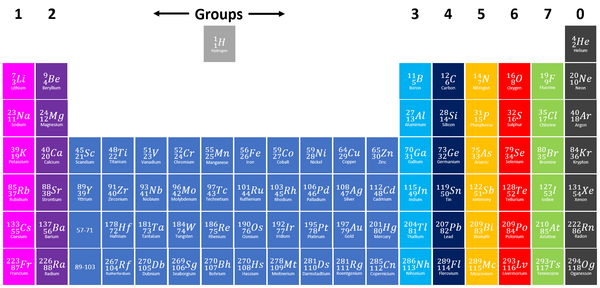
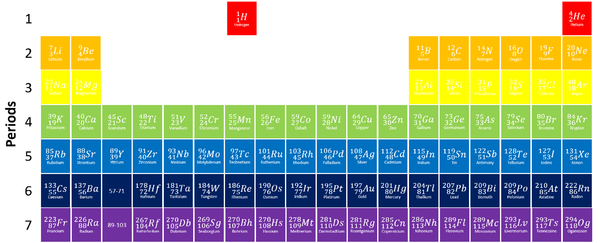
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table is based on an earlier Periodic Table made by a scientist called Mendeleev. Others had tried to arrange all the elements before, but Mendeleev was the first to arrange by both Relative Atomic Mass and chemical properties.
- The modern Periodic Table is only slightly different and is arranged by Atomic Number and chemical properties.
- The columns of the Periodic Table are called groups.
- The rows of the Periodic Table are called periods.
Groups
- The elements are arranged groups of similar chemical properties.
- Elements have similar chemical properties when they have the same number of electrons in the Outer Shell.
Trends within groups
The chemical properties of elements within a group are similar. However, the reactivity within a group changes as you move up or down the periods.
- Group 1: The Alkali Metals all react strongly with water. The reactivity increases as you go down the group.
- Group 2: The Alkali Earth Metals all react strongly with steam and acids. The reactivity increases as you go down the group.
- Group 7: The Halogens all act as bleaching agents and kill bacteria. The reactivity decreases as you go down the group.
- Group 0: The Noble Gases are all inert (unreactive).
The physical properties of elements within a group are similar. However, the property changes gradually as you move down the group.
Periods
- The periods are arranged by the number of Electron Shells.
Trends within Periods
- The chemical and physical properties of elements change as you move along a period.
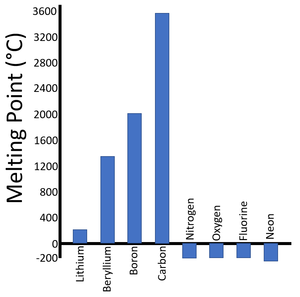
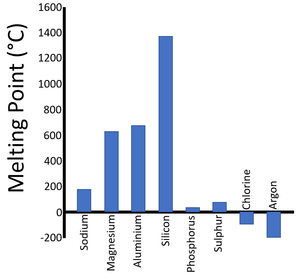
| Period 2 Melting Points | Period 3 Melting Points |
| There is a trend in the Melting Points as you move along the period. | A similar trend can be seen in the next period. |
Key Stage 4
Meaning
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table has the elements arranged by Atomic Number and chemical properties.
- Elements on the Periodic Table are written with their Chemical Symbols and two numbers representing their Relative Atomic Mass and Atomic Number.
- The Relative Atomic Masses on the Periodic Table are an average atomic mass of different isotopes of the same element.
- The columns of the Periodic Table are called groups.
- The rows of the Periodic Table are called periods.