Contents
Key Stage 4
Meaning
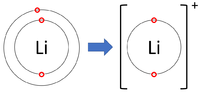
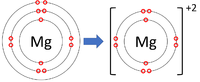
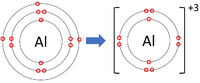
Positive ions are elements which have lost one or more electrons to become positively charged.
About Positive Ions
- In chemical reactions between metals and non-metals the metal elements form positive ions.
- Hydrogen forms positive ions in some compounds and it is these H+ ions which can make solutions acidic.
- Positive ions are attracted to negative ions and to the negative electrode (cathode) during electrolysis.
Examples
| Lithium forms +1 ions. | Magnesium forms +2 ions. | Aluminium forms +3 ions. |
References
AQA
- Positive ion, testing, pages 263, 274-5, GCSE Chemistry; Student Book, Collins, AQA
- Positive ions, page 70, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Positive ions, pages 38, 186-187, 190-191, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Positive ions, pages 72, 258, 259, GCSE Chemistry, CGP, AQA
Edexcel
- Positive ions, page 95, GCSE Physics, Pearson Edexcel
- Positive ions, tests for, pages 196-197, GCSE Chemistry, Pearson, Edexcel