Difference between revisions of "Mercury (Element)"
| Line 38: | Line 38: | ||
: [[Mercury (Element)|Mercury]] is [[sonorous]]. | : [[Mercury (Element)|Mercury]] is [[sonorous]]. | ||
: [[Mercury (Element)|Mercury]] is [[ductile]]. | : [[Mercury (Element)|Mercury]] is [[ductile]]. | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1471851370/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851370&linkCode=as2&tag=nrjc-21&linkId=01c69b0ae058f809cf636033e6ba793e ''Mercury, page 249, GCSE Physics, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158770/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158770&linkCode=as2&tag=nrjc-21&linkId=ec31595e720e1529e49876c3866fff6e ''Mercury, page 58, GCSE Physics; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Mercury, pages 12, 285, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
Revision as of 14:34, 8 November 2019
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
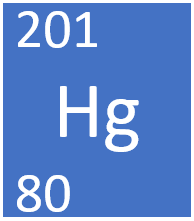
Mercury is a transition metal element, on the Periodic Table, with an atomic number of 80.
About Mercury (Element)|Mercury
Molecular Structure
- Mercury has the chemical symbol Hg.
- Mercury atoms join together in large numbers to form a giant metal molecule.
Atomic Structure
- Mercury as 80 protons and 121 neutrons in its nucleus giving it an Atomic Number of 80 and an atomic mass of 201.
- Mercury is in Period 6 of the Periodic Table because it has 6 electron shells.
Properties
- Mercury is a metal element so it is a good thermal conductor and a good electrical conductor.
- Mercury is a shiny liquid at room temperature.
- Mercury is malleable.
- Mercury is sonorous.
- Mercury is ductile.
Key Stage 4
Meaning
Mercury is a transition metal element, on the Periodic Table, with 80 protons in the nucleus.
About Mercury
Molecular Structure
- Mercury has the chemical formula Hg.
- Mercury atoms join together in a giant metallic structure.
Atomic Structure
- The most stable isotope of Mercury has 121 neutrons in its nucleus giving it an atomic mass of 201.
- Mercury is in Period 6 of the Periodic Table because it has 6 electron shells.
- Mercury loses electrons to form positive metal ions.
Properties
- Mercury forms ionic bonds with non-metals.
- Mercury is a metal element so it is a good thermal conductor and a good electrical conductor.
- Mercury is a shiny liquid at standard temperature and pressure and has a high melting point.
- Mercury is malleable.
- Mercury is sonorous.
- Mercury is ductile.