Difference between revisions of "Sulphur"
(Created page with "==Key Stage 2== ===Meaning=== Sulphur is a yellow solid. ==Key Stage 3== ===Meaning=== File:SulphurSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symb...") |
|||
| (5 intermediate revisions by the same user not shown) | |||
| Line 4: | Line 4: | ||
==Key Stage 3== | ==Key Stage 3== | ||
| + | [[File:SulphurSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Sulphur]].]] | ||
| + | [[File:S-32_WK.PNG|right|200px|thumb|A 2 dimensional representation of a [[Sulphur]] [[atom]] with 16 [[proton]]s and 16 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 16 [[electron]]s orbiting the [[Atomic Nucleus|nucleus]].]] | ||
| + | |||
===Meaning=== | ===Meaning=== | ||
| − | |||
[[Sulphur]] is a [[Group 6]] [[non-metal]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 16. | [[Sulphur]] is a [[Group 6]] [[non-metal]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 16. | ||
| Line 19: | Line 21: | ||
==Key Stage 4== | ==Key Stage 4== | ||
| + | [[File:SKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Sulphur]].]] | ||
| + | [[File:S-32_WK.PNG|right|200px|thumb|A 2 dimensional representation of the [[Bohr Model]] of a [[Sulphur]]-32 [[isotope]] with 16 [[proton]]s and 16 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 2 [[electron]]s in the first [[Electron Orbital|shell]], 8 in the second and 6 in the [[Outer Shell|outer shell]].]] | ||
===Meaning=== | ===Meaning=== | ||
[[Sulphur]] is a [[Group 6]] [[non-metal]] [[element]], on the [[Periodic Table]], with 16 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | [[Sulphur]] is a [[Group 6]] [[non-metal]] [[element]], on the [[Periodic Table]], with 16 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | ||
| Line 26: | Line 30: | ||
====Molecular Structure==== | ====Molecular Structure==== | ||
: The [[Chemical Formula|chemical formula]] for an [[Sulphur]] [[molecule]] is [[Sulphur|S<sub>8</sub>]]. | : The [[Chemical Formula|chemical formula]] for an [[Sulphur]] [[molecule]] is [[Sulphur|S<sub>8</sub>]]. | ||
| − | : [[Sulphur]] forms a [[Covalent Bond|covalent | + | : [[Sulphur]] forms a [[Covalent Bond|covalent bonds]] with other [[Sulphur]] [[atom]]s to produce a [[Simple Covalent Molecule|simple covalent molecule]] made of 8 [[Sulphur]] [[atom]]s. |
| + | |||
====Atomic Structure==== | ====Atomic Structure==== | ||
: The most common [[Stable Isotope|stable isotope]] of [[Sulphur]] has 16 [[neutron]]s in its [[Atomic Nucleus|nucleus]] giving it an [[Relative Atomic Mass|atomic mass]] of 32. | : The most common [[Stable Isotope|stable isotope]] of [[Sulphur]] has 16 [[neutron]]s in its [[Atomic Nucleus|nucleus]] giving it an [[Relative Atomic Mass|atomic mass]] of 32. | ||
Latest revision as of 14:01, 19 February 2021
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Sulphur is a Group 6 non-metal element, on the Periodic Table, with an atomic number of 16.
About Sulphur
- Sulphur has the chemical symbol S.
Molecular Structure
- The chemical formula for a Sulphur molecule is S8.
Atomic Structure
- Sulphur has 16 protons and 16 neutrons in its nucleus giving it an atomic number of 16 and a atomic mass of 32.
- Sulphur is in Period 3 of the Periodic Table because it has 3 electron shells.
Properties
- Sulphur is a yellow solid at room temperature.
Key Stage 4
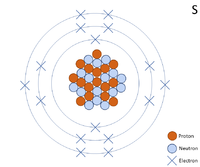
A 2 dimensional representation of the Bohr Model of a Sulphur-32 isotope with 16 protons and 16 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second and 6 in the outer shell.
Meaning
Sulphur is a Group 6 non-metal element, on the Periodic Table, with 16 protons in the nucleus.
About Sulphur
- Sulphur has the chemical symbol S.
Molecular Structure
- The chemical formula for an Sulphur molecule is S8.
- Sulphur forms a covalent bonds with other Sulphur atoms to produce a simple covalent molecule made of 8 Sulphur atoms.
Atomic Structure
- The most common stable isotope of Sulphur has 16 neutrons in its nucleus giving it an atomic mass of 32.
- Sulphur is in Period 3 of the Periodic Table because it has 3 electron shells.
- Sulphur has 6 electrons in its outer shell and needs 2 more to get a full outer shell so it can form 2 bonds with other atoms.
Properties
- Sulphur is yellow solid at standard temperature and pressure with a low melting point.

