Difference between revisions of "Carbon"
| Line 54: | Line 54: | ||
: [[Carbon]] is [[solid]] at [[STP|room temperature]]. | : [[Carbon]] is [[solid]] at [[STP|room temperature]]. | ||
: The different [[allotrope]] have different [[Melting Point|melting points]] and different [[Electrical Conductivity|electrical conductivity]]. | : The different [[allotrope]] have different [[Melting Point|melting points]] and different [[Electrical Conductivity|electrical conductivity]]. | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782945598/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945598&linkCode=as2&tag=nrjc-21&linkId=ad276ad49df77ab4b40ab4fd0fe09738 ''Carbon, page 119, GCSE Combined Science; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158770/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158770&linkCode=as2&tag=nrjc-21&linkId=ec31595e720e1529e49876c3866fff6e ''Carbon, page 287, GCSE Physics; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d66 ''Carbon, page 34, GCSE Chemistry; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Carbon, pages 49-51, 86, 88-89, 196, 223, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
Revision as of 16:47, 2 November 2019
Contents
Key Stage 2
Meaning
Carbon is a chemical that makes diamonds and graphite.
Key Stage 3
Meaning
Carbon is a Group 4 non-metal element, on the Periodic Table, with an atomic number of 6.
About Carbon
- Carbon has the chemical symbol C.
Molecular Structure
Atomic Structure
- Carbon has 6 protons and 6 neutrons in its nucleus giving it an atomic number of 6 and a atomic mass of 12.
- Carbon is in Period 2 of the Periodic Table because it has 2 electron shells.
Properties
- Carbon is solid at room temperature.
Key Stage 4
Meaning
Carbon is a Group 4 non-metal element, on the Periodic Table with 6 protons in the nucleus.
About Carbon
- Carbon has the chemical symbol C.
Molecular Structure
- Carbon forms covalent bonds with other Carbon atoms to produce a giant covalent structure.
- Carbon is able to make long chains of atoms to produce compounds called polymers.
There are several allotropes of Carbon including:
Examples
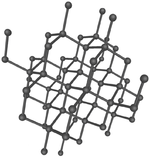
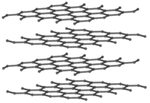
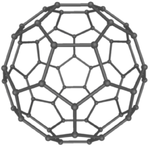
| Diamond is a giant covalent structure where each Carbon atom has 4 bonds with adjacent atoms. | Graphite has a giant covalent structure with each Carbon atom has 3 bonds with adjacent atoms in a layer with loose bonds between the layers. | Graphene has a giant covalent structure where each Carbon atom has 3 bonds with adjacent atoms forming a layer that is one atom thick. | Fullerenes have a giant covalent structure where each Carbon atom has 3 bonds with adjacent atoms forming a sphere. |
Atomic Structure
- The most common isotope of Carbon is Carbon-12 which has 6 protons and 6 neutrons in the nucleus.
- Carbon is in Period 2 of the Periodic Table because it has 2 electron shells.
- Carbon has 4 electrons in its outer shell so it can form 4 bonds with other atoms.
Properties
- Carbon is solid at room temperature.
- The different allotrope have different melting points and different electrical conductivity.