Contents
Key Stage 3
Meaning
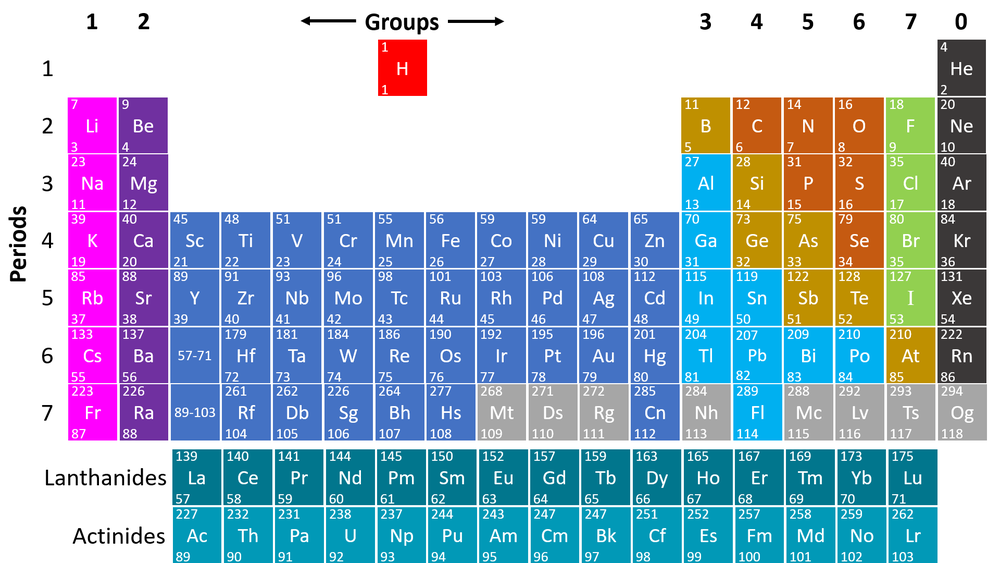
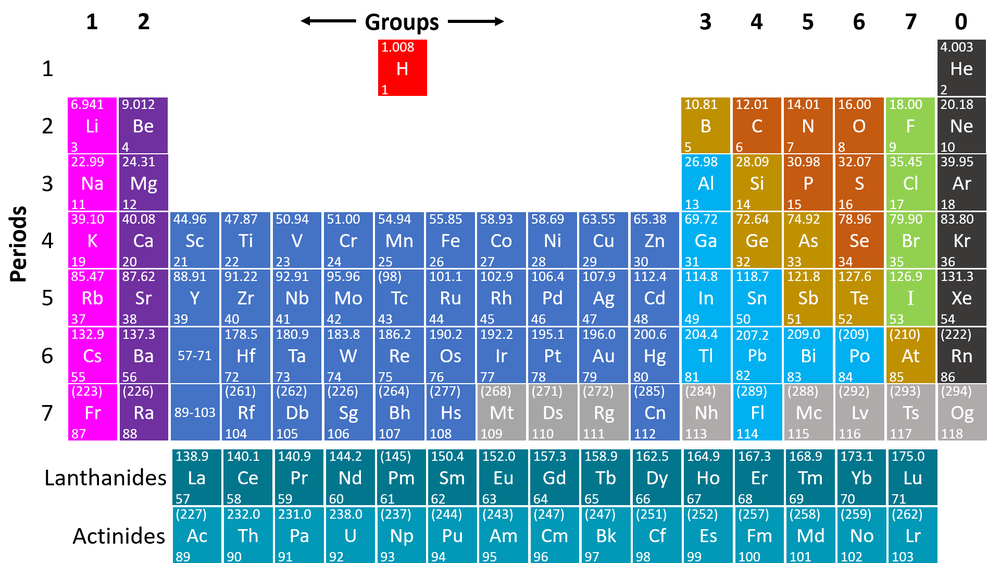
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table is based on an earlier Periodic Table made by a scientist called Mendeleev. Others had tried to arrange all the elements before, but Mendeleev was the first to arrange by both Relative Atomic Mass and chemical properties.
- The modern Periodic Table is only slightly different and is arranged by Atomic Number and chemical properties.
- The columns of the Periodic Table are called groups.
- The rows of the Periodic Table are called periods.
Groups
- The elements are arranged groups of similar chemical properties.
- Elements have similar chemical properties when they have the same number of electrons in the Outer Shell.
Trends within groups
The chemical properties of elements within a group are similar. However, the reactivity within a group changes as you move up or down the periods.
- Group 1: The Alkali Metals all react strongly with water. The reactivity increases as you go down the group.
- Group 2: The Alkali Earth Metals all react strongly with steam and acids. The reactivity increases as you go down the group.
- Group 7: The Halogens all act as bleaching agents and kill bacteria. The reactivity decreases as you go down the group.
- Group 0: The Noble Gases are all inert (unreactive).
The physical properties of elements within a group are similar. However, the property changes gradually as you move down the group.
Periods
- The periods are arranged by the number of Electron Shells.
Trends within Periods
- The chemical and physical properties of elements change as you move along a period.
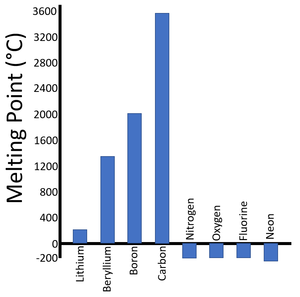
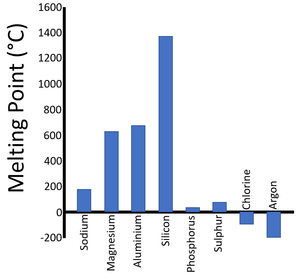
| Period 2 Melting Points | Period 3 Melting Points |
| There is a trend in the Melting Points as you move along the period. | A similar trend can be seen in the next period. |
Key Stage 4
Meaning
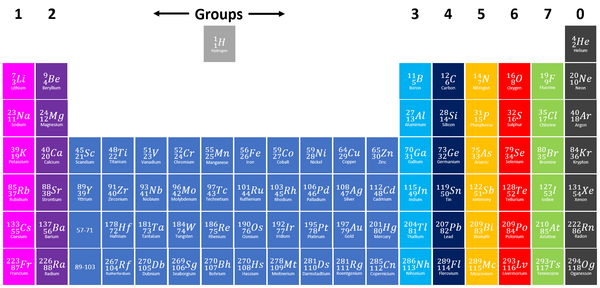
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table has the elements arranged by Atomic Number and chemical properties.
- Elements on the Periodic Table are written with their Chemical Symbols and two numbers representing their Relative Atomic Mass and Atomic Number.
- The Relative Atomic Masses on the Periodic Table are an average atomic mass of different isotopes of the same element.
- The position of an element in the Periodic Table is a result of the Electronic Structure of the element, which itself is due to the Atomic Number of the element.
- The elements in each column of the Periodic Table all have the same number of electrons in the Outer Shell (except Helium in the Nobel Gases) which causes them all to have the same chemical properties. These columns are known as groups.
- The elements in each row of the Periodic Table all have the same number of Electron Shells. These rows are called periods.