Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Sulphur is a Group 6 non-metal element, on the Periodic Table, with an atomic number of 16.
About Sulphur
- Sulphur has the chemical symbol S.
Molecular Structure
- The chemical formula for a Sulphur molecule is S8.
Atomic Structure
- Sulphur has 16 protons and 16 neutrons in its nucleus giving it an atomic number of 16 and a atomic mass of 32.
- Sulphur is in Period 3 of the Periodic Table because it has 3 electron shells.
Properties
- Sulphur is a yellow solid at room temperature.
Key Stage 4
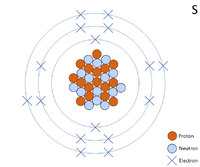
A 2 dimensional representation of the Bohr Model of a Sulphur-32 isotope with 16 protons and 16 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second and 6 in the outer shell.
Meaning
Sulphur is a Group 6 non-metal element, on the Periodic Table, with 16 protons in the nucleus.
About Sulphur
- Sulphur has the chemical symbol S.
Molecular Structure
- The chemical formula for an Sulphur molecule is S8.
- Sulphur forms a covalent bonds with other Sulphur atoms to produce a simple covalent molecule made of 8 Sulphur atoms.
Atomic Structure
- The most common stable isotope of Sulphur has 16 neutrons in its nucleus giving it an atomic mass of 32.
- Sulphur is in Period 3 of the Periodic Table because it has 3 electron shells.
- Sulphur has 6 electrons in its outer shell and needs 2 more to get a full outer shell so it can form 2 bonds with other atoms.
Properties
- Sulphur is yellow solid at standard temperature and pressure with a low melting point.

