Difference between revisions of "Fluorine"
(→Molecular Structure) |
(→Atomic Structure) |
||
| Line 39: | Line 39: | ||
: An [[atom]] of [[Fluorine]] is missing one [[electron]] from having a full [[Outer Shell|outer shell]]. | : An [[atom]] of [[Fluorine]] is missing one [[electron]] from having a full [[Outer Shell|outer shell]]. | ||
: '''Fluoride''' [[ion]]s gain 1 [[electron]] to get a full [[Outer Shell|outer shell]] and become [[Negative Charged|negatively charged]]. | : '''Fluoride''' [[ion]]s gain 1 [[electron]] to get a full [[Outer Shell|outer shell]] and become [[Negative Charged|negatively charged]]. | ||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:FluorineDotandCrossDiagram.png|center|400px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |A [[diagram]] showing the formation of a '''Fluoride''' [[ion]]. | ||
| + | |} | ||
| + | |||
====Properties==== | ====Properties==== | ||
: [[Fluorine]] is the most [[Reactivity|reactive]] [[Halogen]]. | : [[Fluorine]] is the most [[Reactivity|reactive]] [[Halogen]]. | ||
Revision as of 17:20, 31 March 2019
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Fluorine is a Group 7 element, on the Periodic Table, with an atomic number of 9.
About Fluorine
Molecular Structure
- Fluorine has the chemical formula F2.
Atomic Structure
- Fluorine as 9 protons and 10 neutrons in its nucleus giving it an Atomic Number of 9 and an atomic mass of 19.
- An atom of Fluorine is missing one electron from having a full outer shell.
- Fluoride ions gain 1 electron to get a full outer shell and become negatively charged.
Properties
- Fluorine is the most reactive Halogen.
- Fluorine reacts strongly with Hydrogen to produce Hydrogen Fluoride which dissolves in water to produce Hydrofluoric Acid.
- Fluorine is a strong bleaching agent.
- Fluorine kills bacteria.
- Fluorine is a yellow coloured gas at room temperature.
Key Stage 4
Meaning
Fluorine is a Group 2 element, on the Periodic Table, with 9 protons in the nucleus.
About Fluorine
Molecular Structure
- Fluorine has the chemical formula F2.
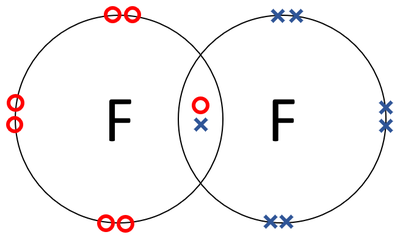
- Fluorine atoms join together in a covalent bond.
| A dot and cross diagram of a Fluorine molecule. |
Atomic Structure
- The most stable isotope of Fluorine has 10 neutrons in its nucleus giving it an atomic mass of 19.
- An atom of Fluorine is missing one electron from having a full outer shell.
- Fluoride ions gain 1 electron to get a full outer shell and become negatively charged.
| A diagram showing the formation of a Fluoride ion. |
Properties
- Fluorine is the most reactive Halogen.
- Fluorine reacts strongly with Hydrogen to produce Hydrogen Fluoride which dissolves in water to produce Hydrofluoric Acid.
- Fluorine is a strong bleaching agent.
- Fluorine kills bacteria.
- Fluorine is a yellow coloured gas at standard temperature and pressure.
Testing For Fluorine
- Collect the gas in a test tube.
- Place a piece of litmus paper over the mouth of the test tube.
- If the litmus paper is bleached white then the gas is Fluorine or Chlorine.