Difference between revisions of "Chlorine"
| (2 intermediate revisions by the same user not shown) | |||
| Line 4: | Line 4: | ||
==Key Stage 3== | ==Key Stage 3== | ||
[[File:ChlorineSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Chlorine]].]] | [[File:ChlorineSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Chlorine]].]] | ||
| − | [[File: | + | [[File:Cl-35_WK.PNG|right|200px|thumb|A 2 dimensional representation of a [[Chlorine]] [[atom]] with 17 [[proton]]s and 18 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 17 [[electron]]s orbiting the [[Atomic Nucleus|nucleus]].]] |
===Meaning=== | ===Meaning=== | ||
[[Chlorine]] is a [[Group 7]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 17. | [[Chlorine]] is a [[Group 7]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 17. | ||
| Line 24: | Line 24: | ||
==Key Stage 4== | ==Key Stage 4== | ||
[[File:ClKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Chlorine]].]] | [[File:ClKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Chlorine]].]] | ||
| + | [[File:Cl-35_WK.PNG|right|200px|thumb|A 2 dimensional representation of the [[Bohr Model]] of a [[Chlorine]]-35 [[isotope]] with 17 [[proton]]s and 18 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 2 [[electron]]s in the first [[Electron Orbital|shell]], 8 in the second and 7 in the [[Outer Shell|outer shell]].]] | ||
===Meaning=== | ===Meaning=== | ||
[[Chlorine]] is a [[Group 7]] [[element]], on the [[Periodic Table]], with 17 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | [[Chlorine]] is a [[Group 7]] [[element]], on the [[Periodic Table]], with 17 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | ||
Latest revision as of 14:01, 19 February 2021
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Chlorine is a Group 7 element, on the Periodic Table, with an atomic number of 17.
About Chlorine
Molecular Structure
- Chlorine has the chemical formula Cl2.
Atomic Structure
- Chlorine as 17 protons and 18 neutrons in its nucleus giving it an Atomic Number of 17 and an atomic mass of 35.
- An atom of Chlorine is missing one electron from having a full outer shell.
Properties
- Chlorine is a non-metal element.
- Chlorine is a more reactive Halogen than Bromine but less reactive than Fluorine.
- Chlorine reacts strongly with Hydrogen to produce Hydrogen Chloride which dissolves in water to produce Hydrochloric Acid.
- Chlorine is a strong bleaching agent.
- Chlorine kills bacteria.
- Chlorine is a pale green coloured gas at room temperature.
Key Stage 4
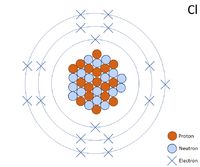
A 2 dimensional representation of the Bohr Model of a Chlorine-35 isotope with 17 protons and 18 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second and 7 in the outer shell.
Meaning
Chlorine is a Group 7 element, on the Periodic Table, with 17 protons in the nucleus.
About Chlorine
Molecular Structure
- Chlorine has the chemical formula Cl2.
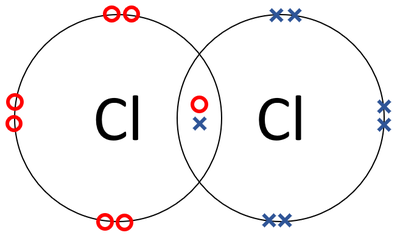
- Chlorine atoms join together in a covalent bond.
| A dot and cross diagram of a Chlorine molecule. |
Atomic Structure
- There are two stable isotopes of Chlorine, one with 18 neutrons and the other with 20 neutrons. They occur in a ratio of roughly 3:1 so the atomic mass of Chlorine is 35.5.
| Chlorine has two common stable Isotopes; Chlorine-35 and Chlorine-37. Chlorine-35 is three times more common than Chlorine-37 so the ratio is 3:1. Find the average atomic mass of Chlorine. |
| 1. State the known quantities.
Ratio: 3:1 3xChlorine-35 : 1xChlorine-37 |
| 2. Substitute the numbers into the equation and solve.
To find the average\[Average=\frac{35+35+35+37}{4}\] \(Average=\frac{35 \times 3 + 37 \times 1}{4}\) \(Average=35.5\) So the Relative Atomic Mass of Chlorine is quoted as 35.5 on the Periodic Table. |
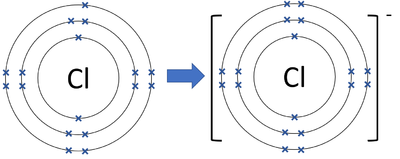
- An atom of Chlorine is missing one electron from having a full outer shell.
- Chloride ions gain 1 electron to get a full outer shell and become negatively charged.
| A diagram showing the formation of a Chloride ion. |
Properties
- Chlorine is a non-metal element.
- Chlorine is a more reactive Halogen than Bromine but less reactive than Fluorine.
- Chlorine reacts strongly with Hydrogen to produce Hydrogen Chloride which dissolves in water to produce Hydrochloric Acid.
- Chlorine is a strong bleaching agent.
- Chlorine kills bacteria.
- Chlorine is a pale green coloured gas at standard temperature and pressure.
Testing For Chlorine
- Collect the gas in a test tube.
- Place a piece of litmus paper over the mouth of the test tube.
- If the litmus paper is bleached white then the gas is Chlorine or Fluorine.
References
AQA
- Chlorine, identification of, page 207, GCSE Chemistry, Hodder, AQA
- Chlorine, page 61, GCSE Chemistry, CGP, AQA
- Chlorine, page 61, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Chlorine, pages 102, 109, 185, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Chlorine, pages 15, 44-5, 64-5, 96, 165, 326, GCSE Chemistry; Student Book, Collins, AQA
- Chlorine, pages 153, 163, GCSE Combined Science; The Revision Guide, CGP, AQA
- Chlorine, pages 88, 102, GCSE Chemistry; The Revision Guide, CGP, AQA
- Chlorine, test for, pages 160, GCSE Combined Science Trilogy 2, Hodder, AQA
- Chlorine; test for, page 203, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Chlorine; test for, page 257, GCSE Chemistry, CGP, AQA
- Chlorine; test, page 273, GCSE Chemistry; Student Book, Collins, AQA