Difference between revisions of "Potassium"
| Line 4: | Line 4: | ||
==Key Stage 3== | ==Key Stage 3== | ||
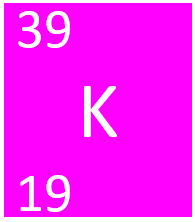
[[File:PotassiumSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Potassium]].]] | [[File:PotassiumSymbol1.png|right|300px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Potassium]].]] | ||
| − | [[File: | + | [[File:K-39_WK.PNG|right|200px|thumb|A 2 dimensional representation of a [[Potassium]] [[atom]] with 19 [[proton]]s and 20 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 19 [[electron]]s orbiting the [[Atomic Nucleus|nucleus]].]] |
===Meaning=== | ===Meaning=== | ||
[[Potassium]] is a [[Group 1]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 19. | [[Potassium]] is a [[Group 1]] [[element]], on the [[Periodic Table]], with an [[Atomic Number|atomic number]] of 19. | ||
| Line 23: | Line 23: | ||
==Key Stage 4== | ==Key Stage 4== | ||
[[File:KKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Potassium]].]] | [[File:KKS4.PNG|right|200px|thumb|The [[Chemical Symbol|chemical symbol]] for [[Potassium]].]] | ||
| − | [[File: | + | [[File:K-39_WK.PNG|right|200px|thumb|A 2 dimensional representation of the [[Bohr Model]] of a [[Potassium]]-39 [[isotope]] with 19 [[proton]]s and 20 [[neutron]]s in the [[Atomic Nucleus|nucleus]] and 2 [[electron]]s in the first [[Electron Orbital|shell]], 8 in the second, 8 in the third and 1 in the [[Outer Shell|outer shell]].]] |
===Meaning=== | ===Meaning=== | ||
[[Potassium]] is a [[Group 1]] [[element]], on the [[Periodic Table]], with 19 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | [[Potassium]] is a [[Group 1]] [[element]], on the [[Periodic Table]], with 19 [[proton]]s in the [[Atomic Nucleus|nucleus]]. | ||
Latest revision as of 14:03, 19 February 2021
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Potassium is a Group 1 element, on the Periodic Table, with an atomic number of 19.
About Potassium
Molecular Structure
- Potassium has the chemical symbol K.
- Potassium atoms join together in large numbers to form a giant metal molecule.
Atomic Structure
- Potassium has 19 protons and 20 neutrons in its nucleus giving it an Atomic Number of 19 and an atomic mass of 39.
- An atom of Potassium has only 1 electron in its outer shell.
Properties
- Potassium is a more reactive alkali metal than Sodium but less reactive than Rubidium.
- Potassium is more reactive than Carbon on the reactivity series so it must be extracted from its ore using electrolysis.
- Potassium oxidises quickly in the presence of Oxygen so it must be stored in oil.
- Potassium reacts strongly with water to produce Hydrogen gas and Potassium Hydroxide.
- Potassium is a solid at room temperature.
Key Stage 4
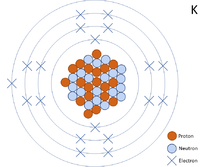
A 2 dimensional representation of the Bohr Model of a Potassium-39 isotope with 19 protons and 20 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second, 8 in the third and 1 in the outer shell.
Meaning
Potassium is a Group 1 element, on the Periodic Table, with 19 protons in the nucleus.
About Potassium
Molecular Structure
- Potassium has the chemical symbol K.
- Potassium atoms join together in a giant metallic structure.
Atomic Structure
- The most stable isotope of Potassium has 20 neutrons in its nucleus giving it an atomic mass of 39.
- An atom of Potassium has only 1 electron in its outer shell.
- Potassium ions have lost an electron to become positively charged.
Properties
- Potassium is a more reactive alkali metal than Sodium but less reactive than Rubidium.
- Potassium is more reactive than Carbon on the reactivity series so it must be extracted from its ore using electrolysis.
- Potassium oxidises quickly in the presence of Oxygen so it must be stored in oil.
- Potassium reacts strongly with water to produce Hydrogen gas and Potassium Hydroxide.
- Potassium is a solid at standard temperature and pressure with a melting point of 63.5 °C.
References
AQA
- Potassium, pages 42-4, 134, 275, 285, 346, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; chloride, pages 44, 71, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; dichromate (VI), page 243, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; hydroxide, pages 148-9, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; iodide, page 115, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; oxide, page 35, GCSE Chemistry; Student Book, Collins, AQA
- Potassium; sulphide, page 91, GCSE Chemistry; Student Book, Collins, AQA
- Potassium-40, page 122, GCSE Physics; Student Book, Collins, AQA